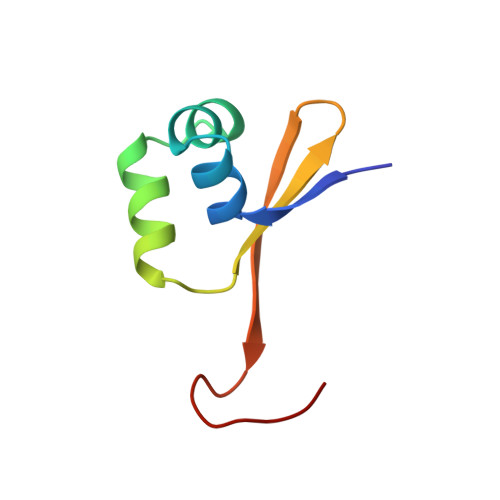
Three-dimensional dimer structure of the lambda-Cro repressor in solution as determined by heteronuclear multidimensional NMR.
Matsuo, H., Shirakawa, M., Kyogoku, Y.(1995) J Mol Biology 254: 668-680
- PubMed: 7500341 Search on PubMed
- DOI: https://doi.org/10.1006/jmbi.1995.0646
- Primary Citation Related Structures:
1COP - PubMed Abstract:
The 1H, 15N and 13C magnetic resonances of the lambda-Cro repressor have been assigned almost completely, mainly through the use of heteronuclear multidimensional NMR methods. Inter-subunit NOEs were distinguished by means of heteronuclear spectral editing technique (13C double half filter technique). Based on the distance and dihedral angle constraints derived from the NMR data, the three-dimensional solution structure of the lambda-Cro repressor in the dimeric form has been calculated by the simulated annealing method. The input for the structure calculations consisted of 1H-1H distance constraints, of which 1536 were intra-subunit and 40 were inter-subunit, and dihedral angle, phi, constraints, which numbered 92. The average root-mean-square deviation (RMSD) for all backbone heavy- atoms of the 20 calculated structures for residues 3 to 59 of the total of 66 amino acid residues in both subunits was 1.57 Angstrum, while the average RMSD for each subunit in the same residue range was 0.66 Angstrum. The subunit is composed of three alpha-helices, residues 7 to 13, 16 to 23 and 27 to 36, and a three-stranded anti-parallel beta-sheet composed of residues 3 to 6, 40 to 44 and 50 to 55. The solution structure of the subunit is essentially the same as that in the crystalline form, but the structure of the dimer form in solution differs from that of the dimer unit in the crystalline form. It is suggested that the solution dimer structure is distorted to fit the recognition helices in the major grooves of DNA on complex formation.
- Institute for Protein Research, Osaka University, Japan.
Organizational Affiliation: