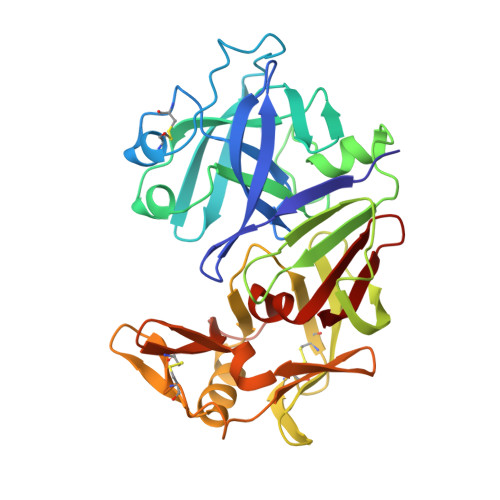
The three-dimensional structure of recombinant bovine chymosin at 2.3 A resolution.
Gilliland, G.L., Winborne, E.L., Nachman, J., Wlodawer, A.(1990) Proteins 8: 82-101
- PubMed: 2217166 Search on PubMed
- DOI: https://doi.org/10.1002/prot.340080110
- Primary Citation Related Structures:
1CMS - PubMed Abstract:
The crystal structure of recombinant bovine chymosin (EC 3.4.23.4; renin), which was cloned and expressed in Escherichia coli, has been determined using X-ray data extending to 2.3 A resolution. The crystals of the enzyme used in this study belong to the space group I222 with unit cell dimensions alpha = 72.7 A, b = 80.3 A, and c = 114.8 A. The structure was solved by the molecular replacement method and was refined by a restrained least-squares procedure. The crystallographic R factor is 0.165 and the deviation of bond distances from ideality is 0.020 A. The resulting model includes all 323 amino acid residues, as well as 297 water molecules. The enzyme has an irregular shape with approximate maximum dimensions of 40 x 50 x 65 A. The secondary structure consists primarily of parallel and antiparallel beta-strands with a few short alpha-helices. The enzyme can be subdivided into N- and C-terminal domains which are separated by a deep cleft containing the active aspartate residues Asp-34 and Asp-216. The amino acid residues and waters at the active site form an extensive hydrogen-bonded network which maintains the pseudo 2-fold symmetry of the entire structure. A comparison of recombinant chymosin with other acid proteinases reveals the high degree of structural similarity with other members of this family of proteins as well as the subtle differences which make chymosin unique. In particular, Tyr-77 of the flap region of chymosin does not hydrogen bond to Trp-42 but protrudes out in the P1 pocket forming hydrophobic interactions with Phe-119 and Leu-32. This may have important implications concerning the mechanism of substrate binding and substrate specificity.
- Center for Advanced Research in Biotechnology, Maryland Biotechnology Institute, University of Maryland, Shady Grove, Rockville 20878.
Organizational Affiliation: