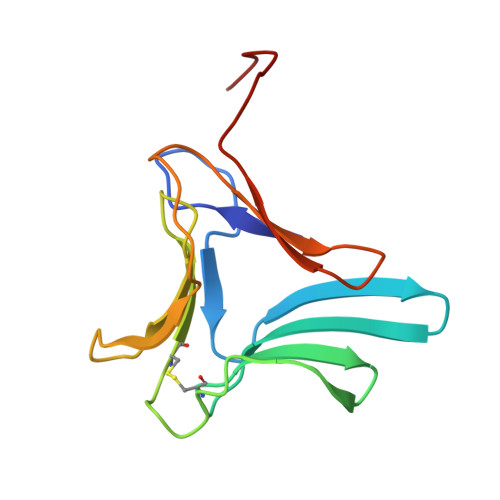
Structure of the native (unligated) mannose-specific bulb lectin from Scilla campanulata (bluebell) at 1.7 A resolution.
Wood, S.D., Wright, L.M., Reynolds, C.D., Rizkallah, P.J., Allen, A.K., Peumans, W.J., Van Damme, E.J.(1999) Acta Crystallogr D Biol Crystallogr 55: 1264-1272
- PubMed: 10393293 Search on PubMed
- DOI: https://doi.org/10.1107/s0907444999005326
- Primary Citation Related Structures:
1B2P - PubMed Abstract:
The X-ray crystal structure of native Scilla campanulata agglutinin, a mannose-specific lectin from bluebell bulbs and a member of the Liliaceae family, has been determined by molecular replacement and refined to an R value of 0.186 at 1.7 A resolution. The lectin crystallizes in space group P21212 with unit-cell parameters a = 70. 42, b = 92.95, c = 46.64 A. The unit cell contains eight protein molecules of Mr = 13143 Da (119 amino-acid residues). The asymmetric unit comprises two chemically identical molecules, A and B, related by a non-crystallographic twofold axis perpendicular to c. This dimer further associates by crystallographic twofold symmetry to form a tetramer. The fold of the polypeptide backbone closely resembles that found in the lectins from Galanthus nivalis (snowdrop) and Hippeastrum (amaryllis) and contains a threefold symmetric beta-prism made up of three antiparallel four-stranded beta-sheets. Each of the four-stranded beta-sheets (I, II and III) possesses a potential saccharide-binding site containing conserved residues; however, site II has two mutations relative to sites I and III which may prevent ligation at this site. Our study provides the first accurate and detailed description of a native (unligated) structure from this superfamily of mannose-specific bulb lectins and will allow comparisons with a number of lectin-saccharide complexes which have already been determined or are currently under investigation.
- School of Biomolecular Sciences, Max Perutz Building, Liverpool John Moores University, Liverpool L3 3AF, England.
Organizational Affiliation: