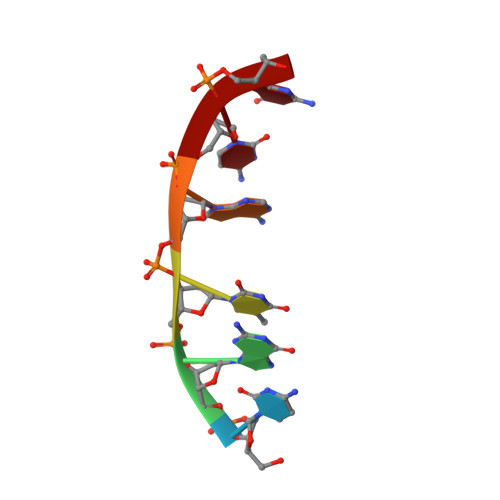
Interactions of deglycosylated cobalt(III)-pepleomycin (green form) with DNA based on NMR structural studies,.
Caceres-Cortes, J., Sugiyama, H., Ikudome, K., Saito, I., Wang, A.H.(1997) Biochemistry 36: 9995-10005
- PubMed: 9254594
- DOI: https://doi.org/10.1021/bi9708951
- Primary Citation Related Structures:
1AO1 - PubMed Abstract:
Pepleomycin (PEP)1 is a metalloglycopeptide antitumor antibiotic that has improved pharmacological properties than does bleomycin (BLM). Both PEP and BLM bind to and degrade DNA in a sequence-selective manner. The binding interactions of HO2--Co(III)-CodPEP (CodPEP) with CGTACG have been studied by 2D NMR and molecular modeling. Inspection of the 2D-NMR data revealed 60 notable intermolecular NOEs between CodPEP and CGTACG which place the drug's metal binding domain and peptide linker in the minor groove of the DNA close to G8 and T9. On the basis of the NOEs, the drug's DNA binding domain is located close to the T9.A4 and A10.T3 base pairs. Intercalation of the bithiazole tail between these base pairs is indicated by the loss of DNA symmetry upon complexation with CodPEP, by a break in the sequential connectivity at the TpA steps, and by the upfield shift of the bithiazole H-H5 and H-H5' proton resonances. Intercalation of the bithiazole moiety unfolds the CodPEP molecule and exposes its hydroperoxide group to the DNA. The hydroperoxide group in the refined model of CodPEP-CGTACG is close to the C4' proton of T9, consistent with cleavage at this position. The NOE pattern between the pyrimidine ring of CodPEP and G8 of DNA suggests a specific pairing recognition via hydrogen bonds between these groups, thus establishing a 5'-GT-3' sequence preference. The structural elucidation of the free CodPEP and CoPEP [Caceres-Cortes et al. (1997) Eur. J. Biochem. 244, 818-828], and of the complex of CodPEP-CGTACG afford a plausible mechanism for the recognition and its subsequent cleaving of DNA by the drug. The process involves the unfolding of the compact CodPEP, recognition of a guanine base using the metal binding domain, threading of the bithiazole tail between base pairs, and finally positioning of the HO2- group close to the T or C found 3' to the specific G site.
- Department of Cell and Structural Biology, University of Illinois at Urbana-Champaign, Urbana, Illinois 61801, USA.
Organizational Affiliation: