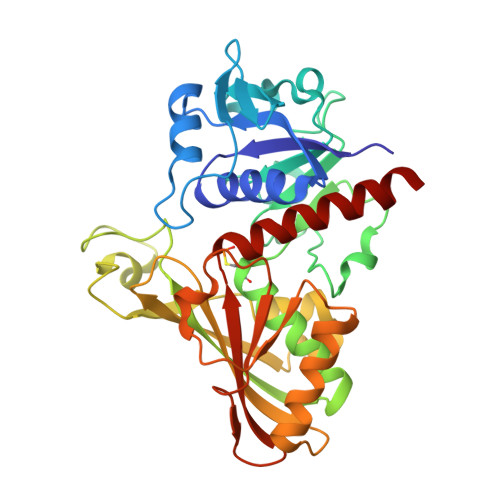
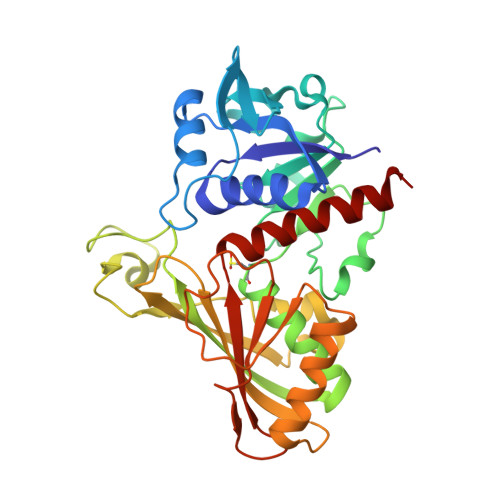
Structure determination and analyses of the GAPDH from the parasite Schistosoma mansoni, the first one from a platyhelminth.
Boreiko, S., Silva, M., Iulek, J.(2021) Biochimie 184: 18-25
- PubMed: 33524435
- DOI: https://doi.org/10.1016/j.biochi.2021.01.014
- Primary Citation of Related Structures:
7JH0 - PubMed Abstract:
The enzyme Glyceraldehyde-3-Phosphate Dehydrogenase from Schistosoma mansoni (SmGAPDH) is characterized as a therapeutical target for schistosomiasis. In this context, we report here the experimental structure, structural analyses and comparisons of SmGAPDH, the first one from a Platyhelminth. The enzyme was expressed, purified and assayed for crystallization, what allowed the obtainment of crystals of sufficient quality to collect X-ray diffraction data up to 2.51 Å resolution. SmGAPDH is the only GAPDH to present the sequence NNR (its residues 114-116) which leads to (especially R116) a hydrogen bond network that possibly reflects on the flexibility of residues to interact with the adenine part of NAD + , speculated to be important for differential drug design.
- Department of Chemistry, State University of Ponta Grossa, Ponta Grossa - PR, 84030-900, Brazil.
Organizational Affiliation: