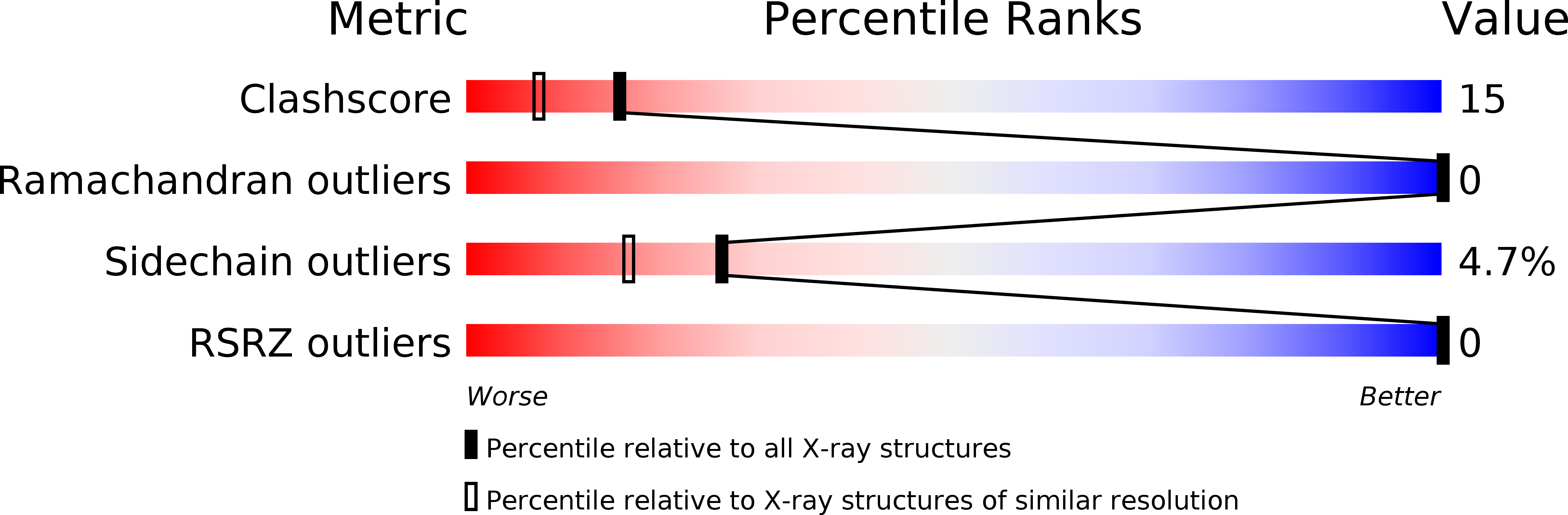Binding mode of azide to ferric Aplysia limacina myoglobin. Crystallographic analysis at 1.9 A resolution.
Mattevi, A., Gatti, G., Coda, A., Rizzi, M., Ascenzi, P., Brunori, M., Bolognesi, M.(1991) J Mol Recognit 4: 1-6
- PubMed: 1931125
- DOI: https://doi.org/10.1002/jmr.300040102
- Primary Citation of Related Structures:
5MBA - PubMed Abstract:
The binding mode of azide to the ferric form of Aplysia limacina myoglobin has been studied by X-ray crystallography. The three-dimensional structure of the complex has been refined at 1.9 A resolution to a crystallographic R-factor of 13.9%, including 126 ordered solvent molecules. Azide binds to the heme iron, at the sixth co-ordination position, and is oriented towards the outer part of the distal site crevice. This orientation is stabilized by an ionic interaction with the side-chain of Arg66 (E10) which, from an outer orientation in the 'aquo-met' ligand-free myoglobin, folds back towards the distal site in the presence of the anionic ligand. In the absence of a hydrogen bond donor residue at the distal E7 position in Aplysia limacina myoglobin, a different polar residue, Arg66 at the E10 topological position, has been selected by molecular evolution in order to grant ligand stabilization.
Organizational Affiliation:
Dipartimento di Genetica e Microbiologia, Università di Pavia, Italy.