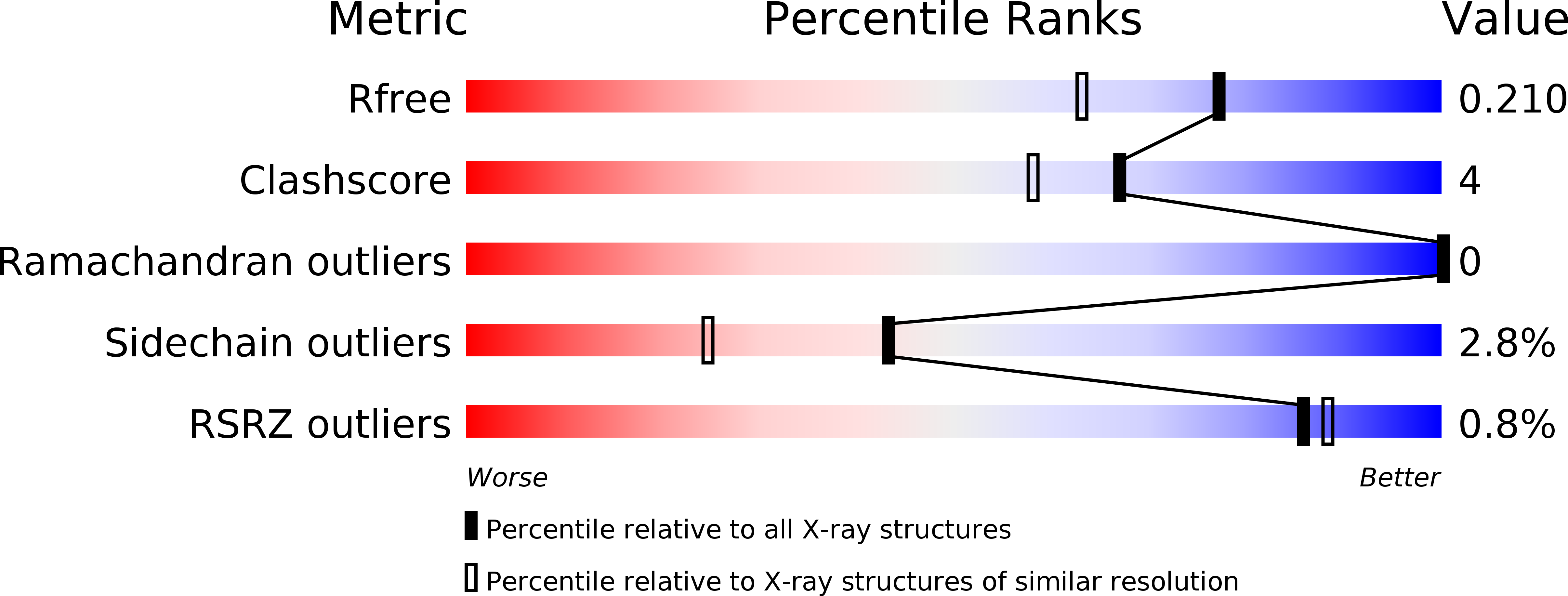
PILR alpha and PILR beta have a siglec fold and provide the basis of binding to sialic acid
Lu, Q., Lu, G., Qi, J., Wang, H., Xuan, Y., Wang, Q., Li, Y., Zhang, Y., Zheng, C., Fan, Z., Yan, J., Gao, G.F.(2014) Proc Natl Acad Sci U S A 111: 8221-8226
- PubMed: 24843130
- DOI: https://doi.org/10.1073/pnas.1320716111
- Primary Citation of Related Structures:
4NFB, 4NFC, 4NFD - PubMed Abstract:
Paired immunoglobulin-like type 2 receptor α (PILRα) and β (PILRβ) belong to the PILR family and are related to innate immune regulation in various species. Despite their high sequence identity, PILRα and PILRβ are shown to have variant sialic acid (SA) binding avidities. To explore the molecular basis of this interaction, we solved the crystal structures of PILRα and PILRβ at resolutions of 1.6 Å and 2.2 Å, respectively. Both molecules adopt a typical siglec fold but use a hydrophobic bond to substitute the siglec-specific disulfide linkage for protein stabilization. We further used HSV-1 glycoprotein B (gB) as a representative molecule to study the PILR-SA interaction. Deploying site-directed mutagenesis, we demonstrated that three residues (Y2, R95, and W108) presented on the surface of PILRα form the SA binding site equivalent to those in siglecs but are arranged in a unique linear mode. PILRβ differs from PILRα in one of these three residues (L108), explaining its inability to engage gB. Mutation of L108 to tryptophan in PILRβ restored the gB-binding capacity. We further solved the structure of this PILRβ mutant complexed with SA, which reveals the atomic details mediating PILR/SA recognition. In comparison with the free PILR structures, amino acid Y2 oriented variantly in the complex structure, thereby disrupting the linear arrangement of PILR residues Y2, R95, and W108. In conclusion, our study provides significant implications for the PILR-SA interaction and paves the way for understanding PILR-related ligand binding.
Organizational Affiliation:
CAS Key Laboratory of Pathogenic Microbiology and Immunology, Institute of Microbiology, Chinese Academy of Sciences, Beijing 100101, China;University of Chinese Academy of Sciences, Beijing 100049, China;