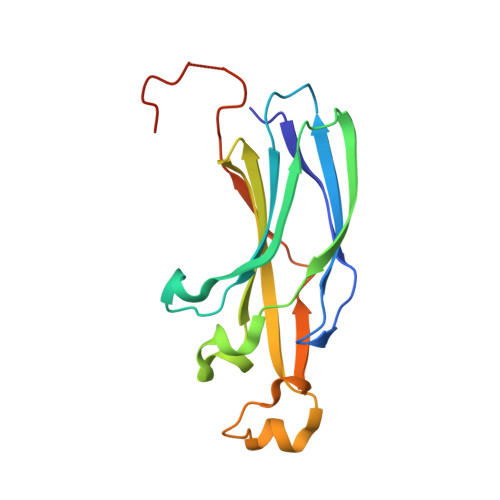
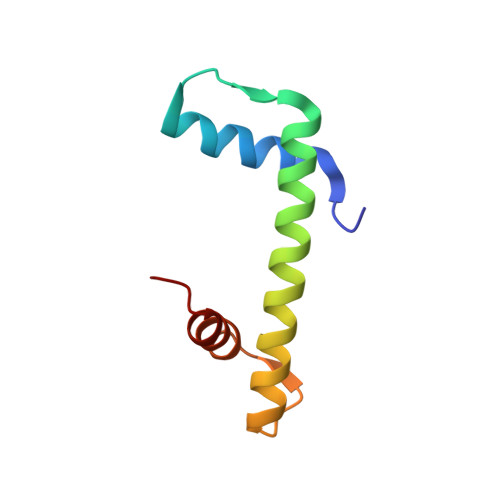
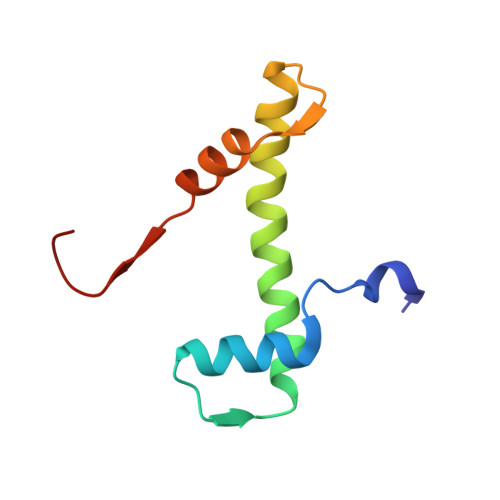
The conformational flexibility of the C-terminus of histone H4 promotes histone octamer and nucleosome stability and yeast viability.
Chavez, M.S., Scorgie, J.K., Dennehey, B.K., Noone, S.M., Tyler, J.K., Churchill, M.E.(2012) Epigenetics Chromatin 5: 5-5
- PubMed: 22541333
- DOI: https://doi.org/10.1186/1756-8935-5-5
- Primary Citation of Related Structures:
4EO5 - PubMed Abstract:
The protein anti-silencing function 1 (Asf1) chaperones histones H3/H4 for assembly into nucleosomes every cell cycle as well as during DNA transcription and repair. Asf1 interacts directly with H4 through the C-terminal tail of H4, which itself interacts with the docking domain of H2A in the nucleosome. The structure of this region of the H4 C-terminus differs greatly in these two contexts.
Organizational Affiliation:
Department of Biochemistry and Molecular Biology, University of Texas MD Anderson Cancer Center, Houston, TX, 77030, USA.