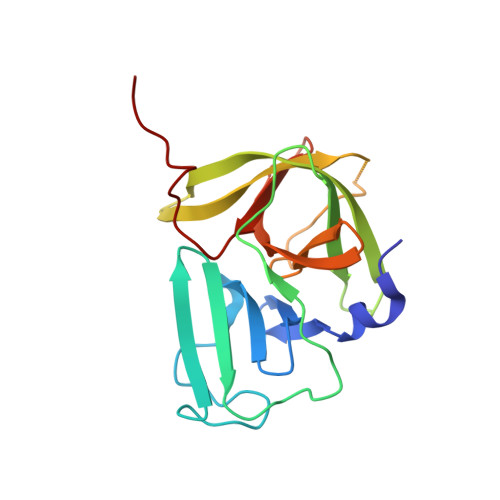
Structure of a Murine Norovirus Ns6 Protease-Product Complex Revealed by Adventitious Crystallisation.
Leen, E.N., Baeza, G., Curry, S.(2012) PLoS One 7: 38723
- PubMed: 22685603
- DOI: https://doi.org/10.1371/journal.pone.0038723
- Primary Citation of Related Structures:
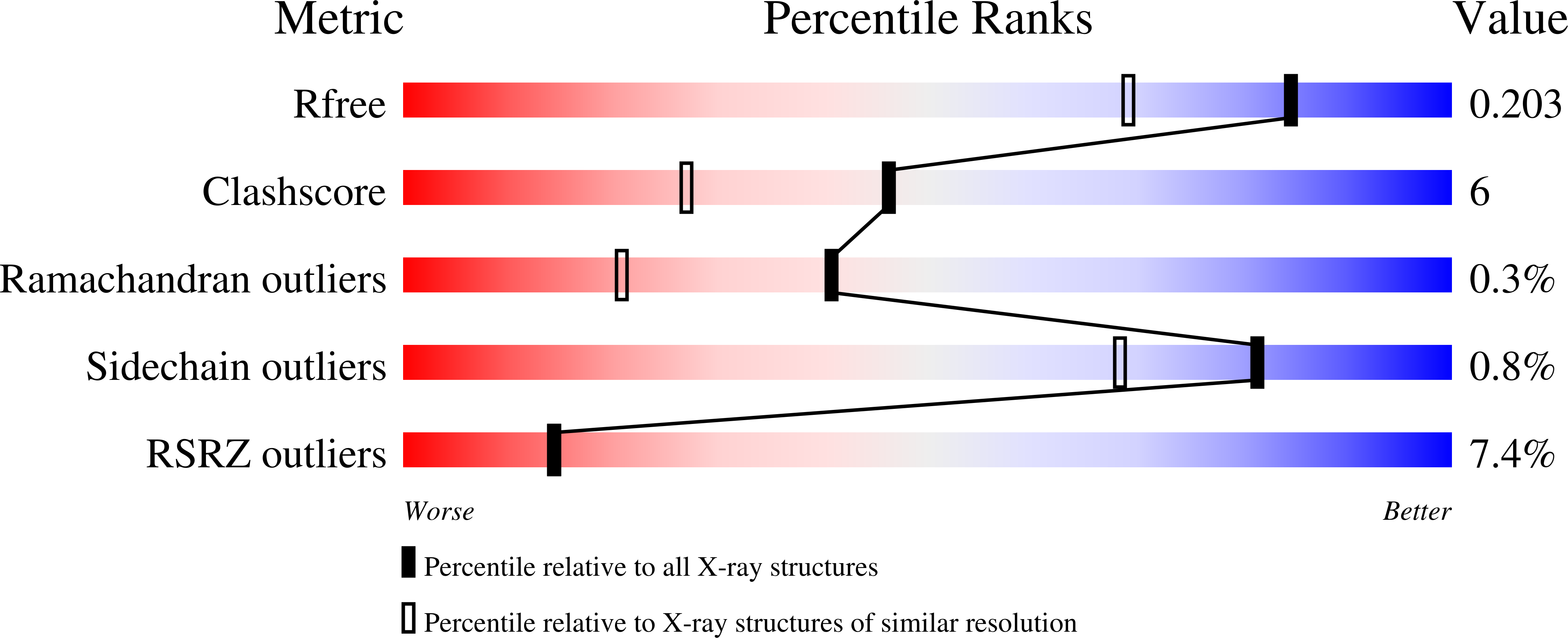
4ASH - PubMed Abstract:
Murine noroviruses have emerged as a valuable tool for investigating the molecular basis of infection and pathogenesis of the closely related human noroviruses, which are the major cause of non-bacterial gastroenteritis. The replication of noroviruses relies on the proteolytic processing of a large polyprotein precursor into six non-structural proteins (NS1-2, NS3, NS4, NS5, NS6(pro), NS7(pol)) by the virally-encoded NS6 protease. We report here the crystal structure of MNV NS6(pro), which has been determined to a resolution of 1.6 Å. Adventitiously, the crystal contacts are mediated in part by the binding of the C-terminus of NS6(pro) within the peptide-binding cleft of a neighbouring molecule. This insertion occurs for both molecules in the asymmetric unit of the crystal in a manner that is consistent with physiologically-relevant binding, thereby providing two independent views of a protease-peptide complex. Since the NS6(pro) C-terminus is formed in vivo by NS6(pro) processing, these crystal contacts replicate the protease-product complex that is formed immediately following cleavage of the peptide bond at the NS6-NS7 junction. The observed mode of binding of the C-terminal product peptide yields new insights into the structural basis of NS6(pro) specificity.
Organizational Affiliation:
Department of Life Sciences, Imperial College, London, United Kingdom.