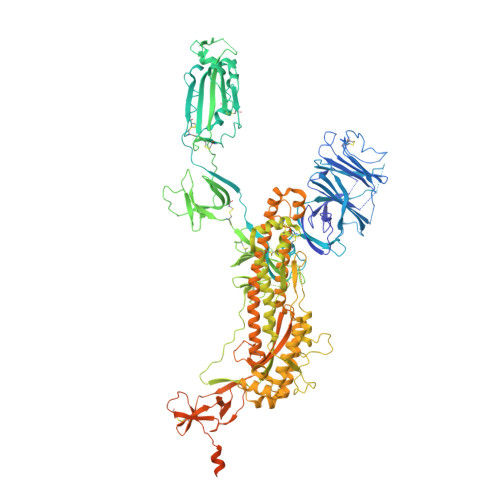
Structure-based design of prefusion-stabilized SARS-CoV-2 spikes.
Hsieh, C.L., Goldsmith, J.A., Schaub, J.M., DiVenere, A.M., Kuo, H.C., Javanmardi, K., Le, K.C., Wrapp, D., Lee, A.G., Liu, Y., Chou, C.W., Byrne, P.O., Hjorth, C.K., Johnson, N.V., Ludes-Meyers, J., Nguyen, A.W., Park, J., Wang, N., Amengor, D., Lavinder, J.J., Ippolito, G.C., Maynard, J.A., Finkelstein, I.J., McLellan, J.S.(2020) Science 369: 1501-1505
- PubMed: 32703906
- DOI: https://doi.org/10.1126/science.abd0826
- Primary Citation of Related Structures:
6XKL - PubMed Abstract:
The coronavirus disease 2019 (COVID-19) pandemic has led to accelerated efforts to develop therapeutics and vaccines. A key target of these efforts is the spike (S) protein, which is metastable and difficult to produce recombinantly. We characterized 100 structure-guided spike designs and identified 26 individual substitutions that increased protein yields and stability. Testing combinations of beneficial substitutions resulted in the identification of HexaPro, a variant with six beneficial proline substitutions exhibiting higher expression than its parental construct (by a factor of 10) as well as the ability to withstand heat stress, storage at room temperature, and three freeze-thaw cycles. A cryo-electron microscopy structure of HexaPro at a resolution of 3.2 angstroms confirmed that it retains the prefusion spike conformation. High-yield production of a stabilized prefusion spike protein will accelerate the development of vaccines and serological diagnostics for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2).
- Department of Molecular Biosciences, University of Texas, Austin, TX 78712, USA.
Organizational Affiliation: