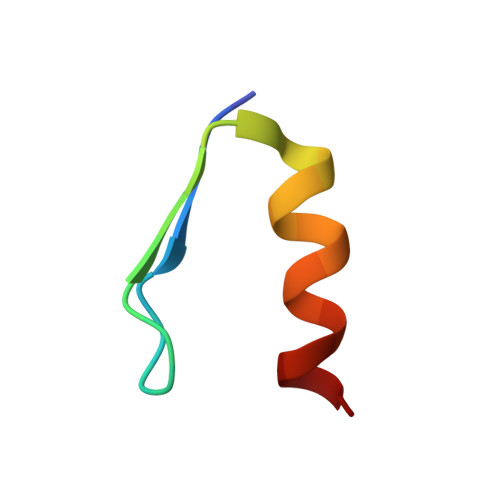
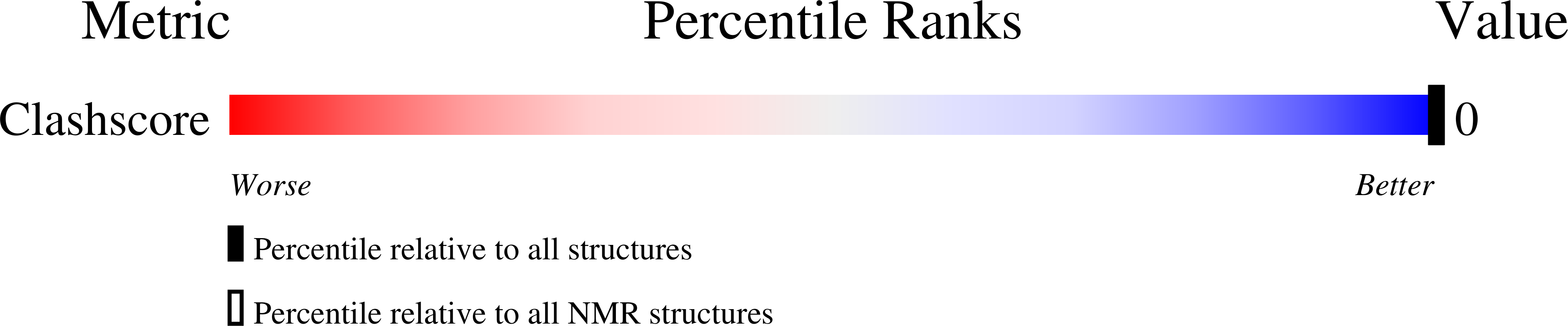Solution structures and characterization of human immunodeficiency virus Rev responsive element IIB RNA targeting zinc finger proteins.
Mishra, S.H., Shelley, C.M., Barrow, D.J., Darby, M.K., Germann, M.W.(2006) Biopolymers 83: 352-364
- PubMed: 16826557
- DOI: https://doi.org/10.1002/bip.20565
- Primary Citation of Related Structures:
2AB3, 2AB7 - PubMed Abstract:
The Rev responsive element (RRE), a part of unspliced human immunodeficiency virus (HIV) RNA, serves a crucial role in the production of infectious HIV virions. The viral protein Rev binds to RRE and facilitates transport of mRNA to the cytoplasm. Inhibition of the Rev-RRE interaction disrupts the viral life cycle. Using a phage display protocol, dual zinc finger proteins (ZNFs) were generated that bind specifically to RREIIB at the high affinity Rev binding site. These proteins were further shortened and simplified, and they still retained their RNA binding affinity. The solution structures of ZNF29 and a mutant, ZNF29G29R, have been determined by nuclear magnetic resonance (NMR) spectroscopy. Both proteins form C(2)H(2)-type zinc fingers with essentially identical structures. RNA protein interactions were evaluated quantitatively by isothermal titration calorimetry, which revealed dissociation constants (K(d)'s) in the nanomolar range. The interaction with the RNA is dependent upon the zinc finger structure; in the presence of EDTA, RNA binding is abolished. For both proteins, RNA binding is mediated by the alpha-helical portion of the zinc fingers and target the bulge region of RREIIB-TR. However, ZNF29G29R exhibits significantly stronger binding to the RNA target than ZNF29; this illustrates that the binding of the zinc finger scaffold is amenable to further improvements.
Organizational Affiliation:
Department of Chemistry, Georgia State University, Atlanta, 30303, USA.