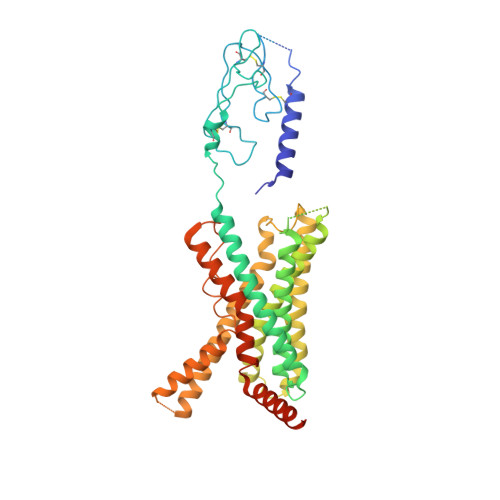
Molecular recognition of two endogenous hormones by the human parathyroid hormone receptor-1.
Zhao, L.H., Yuan, Q.N., Dai, A.T., He, X.H., Chen, C.W., Zhang, C., Xu, Y.W., Zhou, Y., Wang, M.W., Yang, D.H., Xu, H.E.(2023) Acta Pharmacol Sin 44: 1227-1237
- PubMed: 36482086
- DOI: https://doi.org/10.1038/s41401-022-01032-z
- Primary Citation of Related Structures:
8HA0, 8HAF, 8HAO - PubMed Abstract:
Parathyroid hormone (PTH) and PTH-related peptide (PTHrP) are two endogenous hormones recognized by PTH receptor-1 (PTH1R), a member of class B G protein- coupled receptors (GPCRs). Both PTH and PTHrP analogs including teriparatide and abaloparatide are approved drugs for osteoporosis, but they exhibit distinct pharmacology. Here we report two cryo-EM structures of human PTH1R bound to PTH and PTHrP in the G protein-bound state at resolutions of 2.62 Å and 3.25 Å, respectively. Detailed analysis of these structures uncovers both common and unique features for the agonism of PTH and PTHrP. Molecular dynamics (MD) simulation together with site-directed mutagenesis studies reveal the molecular basis of endogenous hormones recognition specificity and selectivity to PTH1R. These results provide a rational template for the clinical use of PTH and PTHrP analogs as an anabolic therapy for osteoporosis and other disorders.
Organizational Affiliation:
The CAS Key Laboratory of Receptor Research, Center for Structure and Function of Drug Targets, Shanghai Institute of Materia Medica, Chinese Academy of Sciences, Shanghai, 201203, China. zhaolihuawendy@simm.ac.cn.