C3- and C3/C5-Epimerases Required for the Biosynthesis of the Capsular Polysaccharides from Campylobacter jejuni .
Ghosh, M.K., Xiang, D.F., Thoden, J.B., Holden, H.M., Raushel, F.M.(2022) Biochemistry 61: 2036-2048
- PubMed: 36093987
- DOI: https://doi.org/10.1021/acs.biochem.2c00364
- Primary Citation of Related Structures:
8DAK, 8DB5, 8DCL, 8DCO - PubMed Abstract:
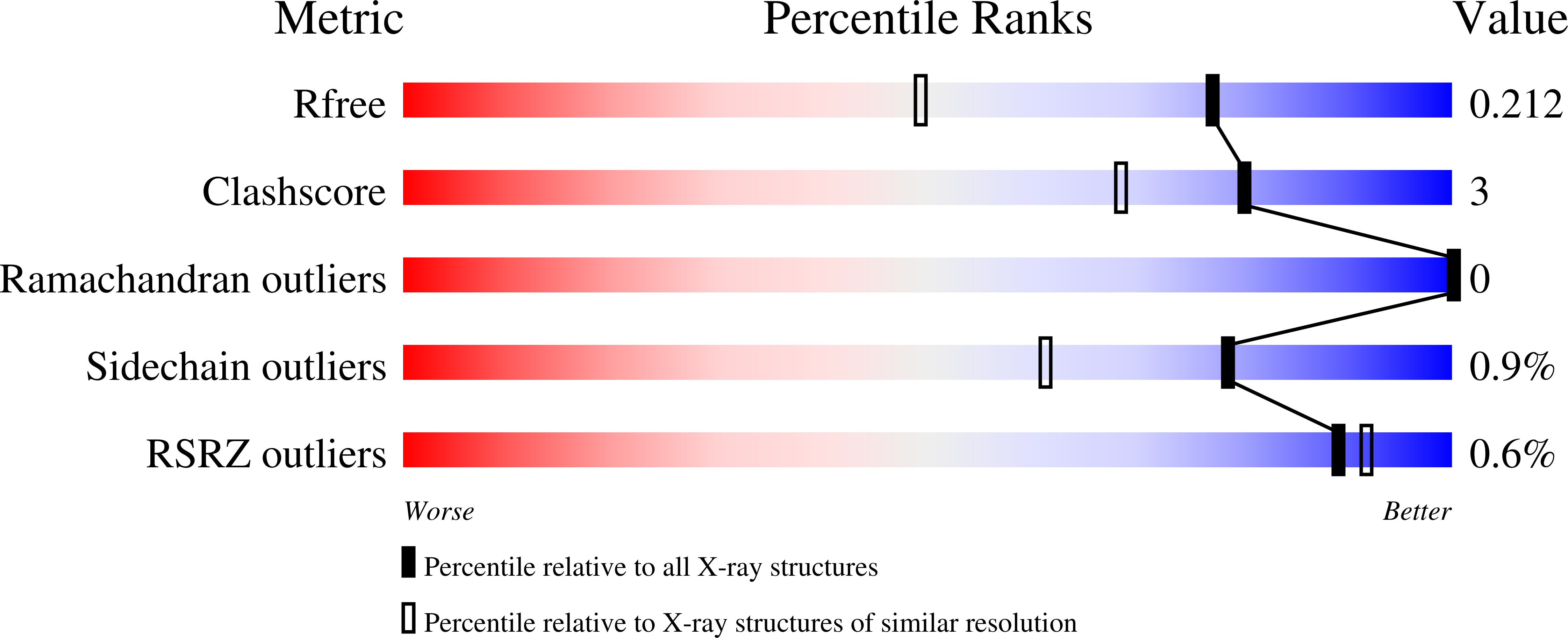
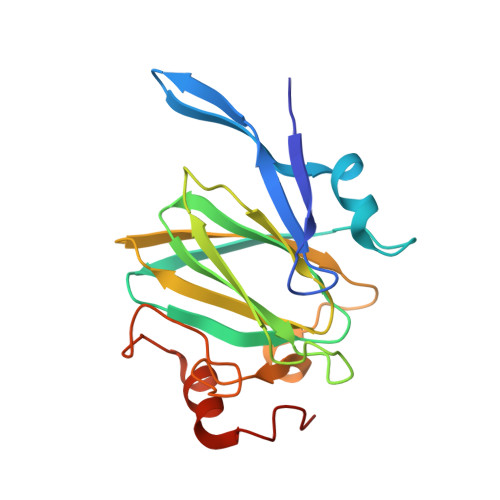
Campylobacter jejuni is a human pathogen and one of the leading causes of food poisoning in Europe and the United States. The outside of the bacterium is coated with a capsular polysaccharide that assists in the evasion of the host immune system. Many of the serotyped strains of C. jejuni contain a 6-deoxy-heptose moiety that is biosynthesized from GDP-d- glycero -d- manno -heptose by the successive actions of a 4,6-dehydratase, a C3/C5-epimerase, and a C4-reductase. We identified 18 different C3/C5-epimerases that could be clustered together into three groups at a sequence identity of >89%. Four of the enzymes from the largest cluster (from serotypes HS:3, HS:10, HS:23/36, and HS:41) were shown to only catalyze the epimerization at C3. Three enzymes from the second largest cluster (HS:2, HS:15, and HS:42) were shown to catalyze the epimerization at C3 and C5. Enzymes from the third cluster were not characterized. The three-dimensional structures of the epimerases from serotypes HS:3, HS:23/36, HS:15, and HS:41 were determined to resolutions of 1.5-1.9 Å. The overall subunit architecture places these enzymes into the diverse "cupin" superfamily. Within X-ray coordinate error, the immediate regions surrounding the active sites are identical, suggesting that factors extending farther out may influence product outcome. The X-ray crystal structures are consistent with His-67 and Tyr-134 acting as general acid/base catalysts for the epimerization of C3 and/or C5. Two amino acid changes (A76V/C136L) were enough to convert the C3-epimerase from serotype HS:3 to one that could now catalyze the epimerization at both C3 and C5.
Organizational Affiliation:
Department of Chemistry, Texas A&M University, College Station, Texas 77843, United States.