An engineered PET hydrolase for biodegradation of microplastics in ocean water
Xie, W., Jia, Q.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Poly(ethylene terephthalate) hydrolase | 270 | Piscinibacter sakaiensis | Mutation(s): 9 Gene Names: ISF6_4831 EC: 3.1.1.101 | 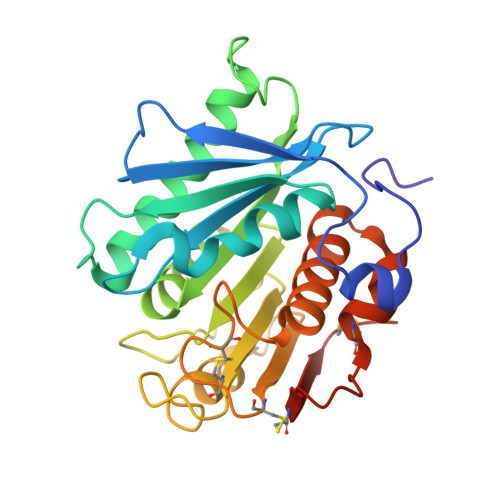 | |
UniProt | |||||
Find proteins for A0A0K8P6T7 (Piscinibacter sakaiensis) Explore A0A0K8P6T7 Go to UniProtKB: A0A0K8P6T7 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A0A0K8P6T7 | ||||
Sequence AnnotationsExpand | |||||
| |||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| CL (Subject of Investigation/LOI) Query on CL | B [auth A], C [auth A], D [auth A] | CHLORIDE ION Cl VEXZGXHMUGYJMC-UHFFFAOYSA-M |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 115.588 | α = 90 |
| b = 50.536 | β = 92.458 |
| c = 41.37 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| HKL-3000 | data reduction |
| HKL-3000 | data scaling |
| PHENIX | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Natural Science Foundation of China (NSFC) | China | 31770075 |
| National Natural Science Foundation of China (NSFC) | China | 31870782 |