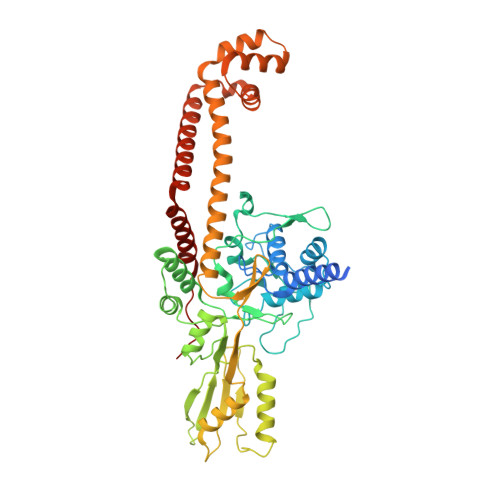
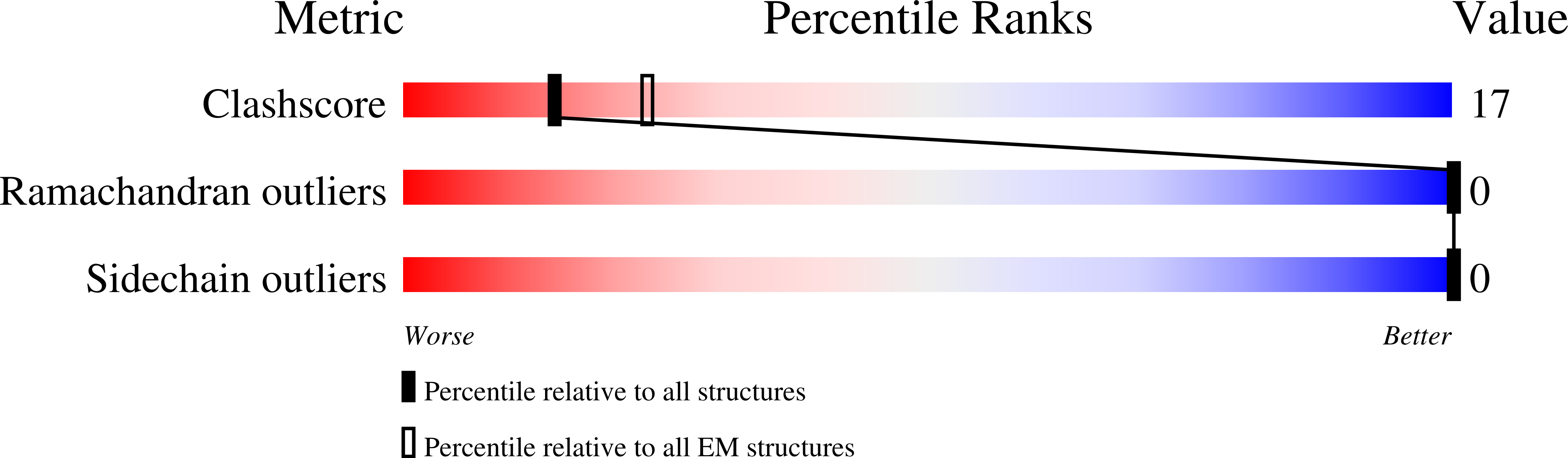CryoEM structures of open dimers of Gyrase A in complex with DNA illuminate mechanism of strand passage.
Soczek, K.M., Grant, T., Rosenthal, P.B., Mondragon, A.(2018) Elife 7
- PubMed: 30457554
- DOI: https://doi.org/10.7554/eLife.41215
- Primary Citation of Related Structures:
6N1P, 6N1Q, 6N1R - PubMed Abstract:
Gyrase is a unique type IIA topoisomerase that uses ATP hydrolysis to maintain the negatively supercoiled state of bacterial DNA. In order to perform its function, gyrase undergoes a sequence of conformational changes that consist of concerted gate openings, DNA cleavage, and DNA strand passage events. Structures where the transported DNA molecule (T-segment) is trapped by the A subunit have not been observed. Here we present the cryoEM structures of two oligomeric complexes of open gyrase A dimers and DNA. The protein subunits in these complexes were solved to 4 Å and 5.2 Å resolution. One of the complexes traps a linear DNA molecule, a putative T-segment, which interacts with the open gyrase A dimers in two states, representing steps either prior to or after passage through the DNA-gate. The structures locate the T-segment in important intermediate conformations of the catalytic cycle and provide insights into gyrase-DNA interactions and mechanism.
Organizational Affiliation:
Department of Molecular Biosciences, Northwestern University, Evanston, United States.