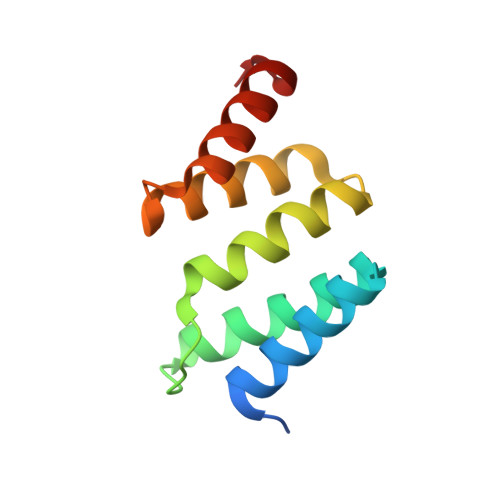
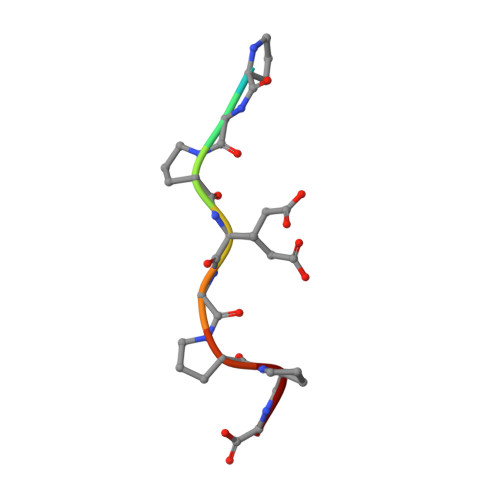
Structural enzymology binding studies of the peptide-substrate-binding domain of human collagen prolyl 4-hydroxylase (type-II): High affinity peptides have a PxGP sequence motif.
Murthy, A.V., Sulu, R., Koski, M.K., Tu, H., Anantharajan, J., Sah-Teli, S.K., Myllyharju, J., Wierenga, R.K.(2018) Protein Sci 27: 1692-1703
- PubMed: 30168208
- DOI: https://doi.org/10.1002/pro.3450
- Primary Citation of Related Structures:
6EVL, 6EVM, 6EVN, 6EVO, 6EVP - PubMed Abstract:
The peptide-substrate-binding (PSB) domain of collagen prolyl 4-hydroxylase (C-P4H, an α 2 β 2 tetramer) binds proline-rich procollagen peptides. This helical domain (the middle domain of the α subunit) has an important role concerning the substrate binding properties of C-P4H, although it is not known how the PSB domain influences the hydroxylation properties of the catalytic domain (the C-terminal domain of the α subunit). The crystal structures of the PSB domain of the human C-P4H isoform II (PSB-II) complexed with and without various short proline-rich peptides are described. The comparison with the previously determined PSB-I peptide complex structures shows that the C-P4H-I substrate peptide (PPG) 3 , has at most very weak affinity for PSB-II, although it binds with high affinity to PSB-I. The replacement of the middle PPG triplet of (PPG) 3 to the nonhydroxylatable PAG, PRG, or PEG triplet, increases greatly the affinity of PSB-II for these peptides, leading to a deeper mode of binding, as compared to the previously determined PSB-I peptide complexes. In these PSB-II complexes, the two peptidyl prolines of its central P(A/R/E)GP region bind in the Pro5 and Pro8 binding pockets of the PSB peptide-binding groove, and direct hydrogen bonds are formed between the peptide and the side chains of the highly conserved residues Tyr158, Arg223, and Asn227, replacing water mediated interactions in the corresponding PSB-I complex. These results suggest that PxGP (where x is not a proline) is the common motif of proline-rich peptide sequences that bind with high affinity to PSB-II.
Organizational Affiliation:
Biocenter Oulu and Faculty of Biochemistry and Molecular Medicine, University of Oulu, Oulu, Finland.