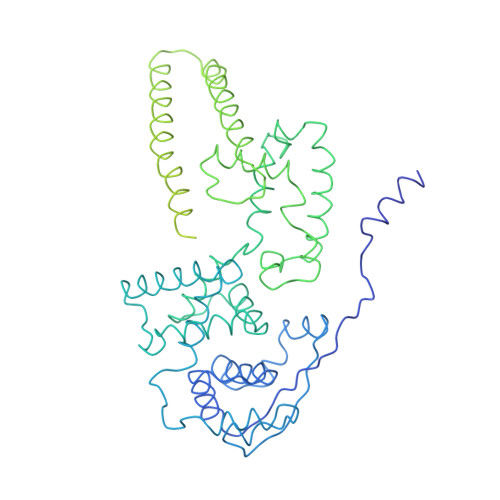
Structure and assembly of the Ebola virus nucleocapsid.
Wan, W., Kolesnikova, L., Clarke, M., Koehler, A., Noda, T., Becker, S., Briggs, J.A.G.(2017) Nature 551: 394-397
- PubMed: 29144446
- DOI: https://doi.org/10.1038/nature24490
- Primary Citation of Related Structures:
6EHL, 6EHM - PubMed Abstract:
Ebola and Marburg viruses are filoviruses: filamentous, enveloped viruses that cause haemorrhagic fever. Filoviruses are within the order Mononegavirales, which also includes rabies virus, measles virus, and respiratory syncytial virus. Mononegaviruses have non-segmented, single-stranded negative-sense RNA genomes that are encapsidated by nucleoprotein and other viral proteins to form a helical nucleocapsid. The nucleocapsid acts as a scaffold for virus assembly and as a template for genome transcription and replication. Insights into nucleoprotein-nucleoprotein interactions have been derived from structural studies of oligomerized, RNA-encapsidating nucleoprotein, and cryo-electron microscopy of nucleocapsid or nucleocapsid-like structures. There have been no high-resolution reconstructions of complete mononegavirus nucleocapsids. Here we apply cryo-electron tomography and subtomogram averaging to determine the structure of Ebola virus nucleocapsid within intact viruses and recombinant nucleocapsid-like assemblies. These structures reveal the identity and arrangement of the nucleocapsid components, and suggest that the formation of an extended α-helix from the disordered carboxy-terminal region of nucleoprotein-core links nucleoprotein oligomerization, nucleocapsid condensation, RNA encapsidation, and accessory protein recruitment.
Organizational Affiliation:
Structural and Computational Biology Unit, European Molecular Biology Laboratory, Meyerhofstraße 1, 69117 Heidelberg, Germany.