The structure and catalytic mechanism of human sphingomyelin phosphodiesterase like 3a - an acid sphingomyelinase homologue with a novel nucleotide hydrolase activity.
Lim, S.M., Yeung, K., Tresaugues, L., Ling, T.H., Nordlund, P.(2016) FEBS J 283: 1107-1123
- PubMed: 26783088
- DOI: https://doi.org/10.1111/febs.13655
- Primary Citation of Related Structures:
2XS6, 5EBB, 5EBE - PubMed Abstract:
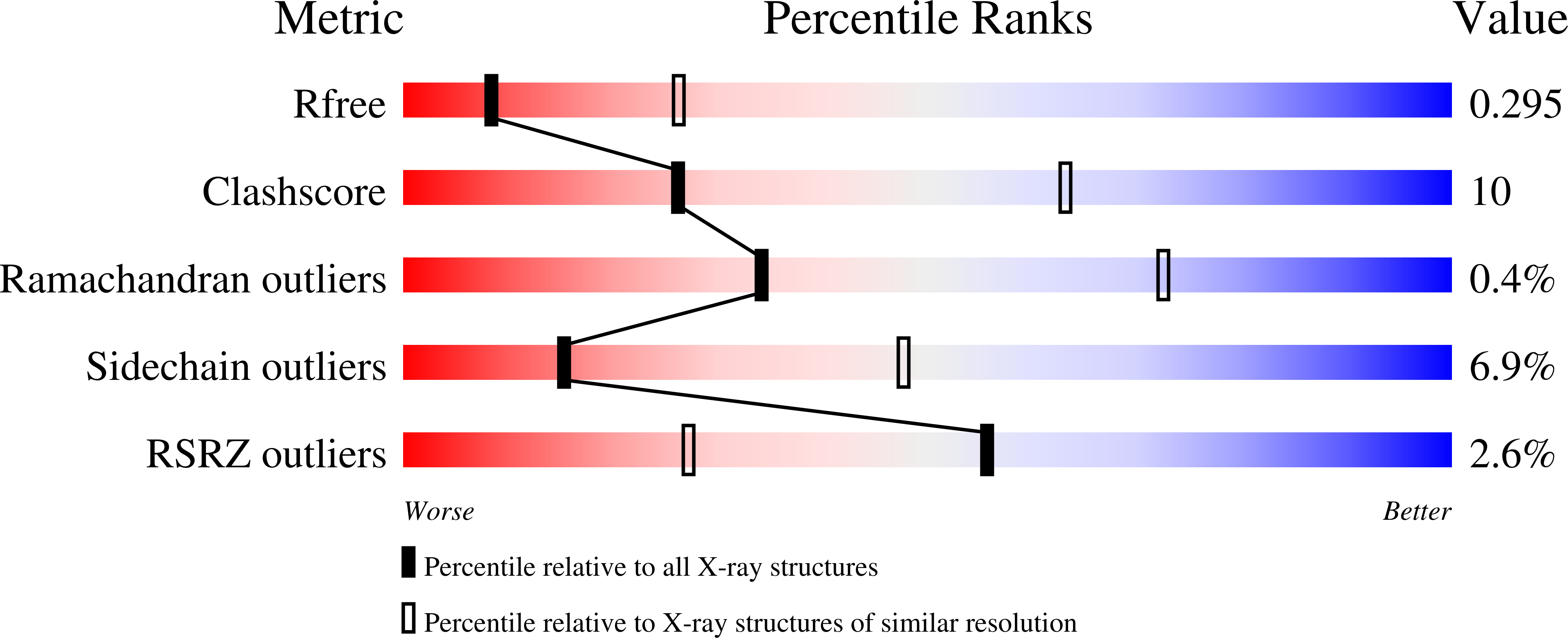
Human sphingomyelinase phosphodiesterase like 3a (SMPDL3a) is a secreted enzyme that shares a conserved catalytic domain with human acid sphingomyelinase (aSMase), the enzyme carrying mutations causative of Niemann-Pick disease. We have solved the structure of SMPDL3a revealing a calcineurin-like fold. A dimetal site, glycosylation pattern and a disulfide bond network are likely to be conserved also in human aSMase. We show that the binuclear site of SMPDL3a is occupied by two Zn(2+) ions and that excess Zn(2+) leads to inhibition of enzyme activity through binding to additional sites. As an extension of recent biochemical work we uncovered that SMPDL3a catalyses the hydrolysis of several modified nucleotides that include cytidine 5'-diphosphocholine, cytidine diphosphate ethanolamine and ADP-ribose, but not the aSMase substrate, sphingomyelin. We subsequently determined the structure of SMPDL3a in complex with the product 5'-cytidine monophosphate (CMP), a structure that is consistent with several distinct coordination modes of the substrate/product in the active site during the reaction cycle. Based on the structure of CMP complexes, we propose a phosphoryl transfer mechanism for SMPDL3a. Finally, a homology model of human aSMase was constructed to allow for the mapping of selected Niemann-Pick disease mutations on a three-dimensional framework to guide further characterization of their effects on aSMase function.
Organizational Affiliation:
Division of Biomedical Structural Biology, School of Biological Sciences, Nanyang Technological University, Singapore.