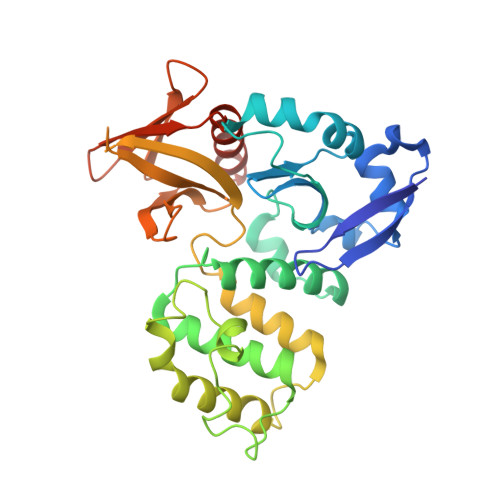
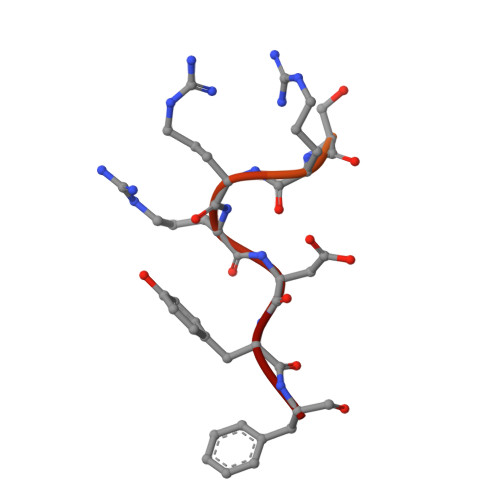
Structural basis of the junctional anchorage of the cerebral cavernous malformations complex.
Gingras, A.R., Liu, J.J., Ginsberg, M.H.(2012) J Cell Biol 199: 39-48
- PubMed: 23007647
- DOI: https://doi.org/10.1083/jcb.201205109
- Primary Citation of Related Structures:
3U7D - PubMed Abstract:
The products of genes that cause cerebral cavernous malformations (CCM1/KRIT1, CCM2, and CCM3) physically interact. CCM1/KRIT1 links this complex to endothelial cell (EC) junctions and maintains junctional integrity in part by inhibiting RhoA. Heart of glass (HEG1), a transmembrane protein, associates with KRIT1. In this paper, we show that the KRIT1 band 4.1, ezrin, radixin, and moesin (FERM) domain bound the HEG1 C terminus (K(d) = 1.2 µM) and solved the structure of this assembly. The KRIT1 F1 and F3 subdomain interface formed a hydrophobic groove that binds HEG1(Tyr(1,380)-Phe(1,381)), thus defining a new mode of FERM domain-membrane protein interaction. This structure enabled design of KRIT1(L717,721A), which exhibited a >100-fold reduction in HEG1 affinity. Although well folded and expressed, KRIT1(L717,721A) failed to target to EC junctions or complement the effects of KRIT1 depletion on zebrafish cardiovascular development or Rho kinase activation in EC. These data establish that this novel FERM-membrane protein interaction anchors CCM1/KRIT1 at EC junctions to support cardiovascular development.
Organizational Affiliation:
Department of Medicine, University of California, San Diego, La Jolla, CA 92093, USA.