Transglutaminase 2 in complex with a novel inhibitor
Lindemann, I., Boettcher, J., Oertel, K., Weber, J., Hils, M., Pasternack, R., Heine, A., Klebe, G.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Protein-glutamine gamma-glutamyltransferase 2 | 694 | Homo sapiens | Mutation(s): 0 Gene Names: TGM2 EC: 2.3.2.13 | 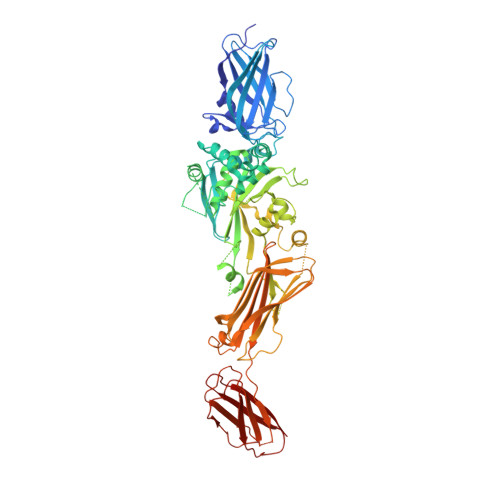 | |
UniProt & NIH Common Fund Data Resources | |||||
Find proteins for P21980 (Homo sapiens) Explore P21980 Go to UniProtKB: P21980 | |||||
PHAROS: P21980 GTEx: ENSG00000198959 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P21980 | ||||
Sequence AnnotationsExpand | |||||
| |||||
Find similar proteins by: Sequence | 3D Structure
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Peptide inhibitor | 5 | synthetic construct | Mutation(s): 0 | 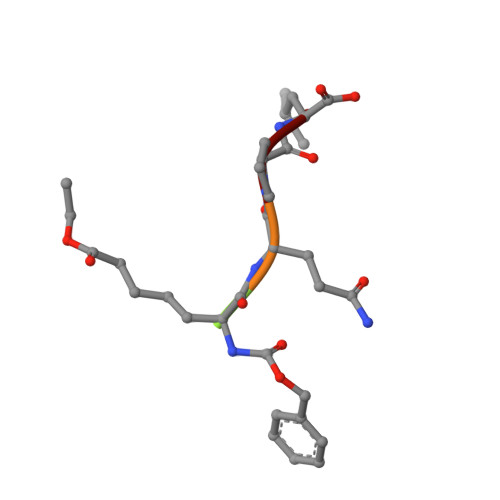 | |
Sequence AnnotationsExpand | |||||
| |||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| SO4 Query on SO4 | C [auth A] D [auth A] E [auth A] F [auth A] G [auth A] | SULFATE ION O4 S QAOWNCQODCNURD-UHFFFAOYSA-L |  | ||
| Modified Residues 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| XW1 Query on XW1 | B | L-PEPTIDE LINKING | C9 H17 N O4 |  | ALA |
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| ID | Chains | Name | Type/Class | 2D Diagram | 3D Interactions |
| PRD_001095 Query on PRD_001095 | B | N~2~-[(2S)-2-{[(benzyloxy)carbonyl]amino}-7-ethoxy-7-oxoheptanoyl]-L-glutaminyl-L-prolyl-L-leucine | Peptide-like / Inhibitor |  | |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 71 | α = 90 |
| b = 71 | β = 90 |
| c = 310.36 | γ = 90 |
| Software Name | Purpose |
|---|---|
| MAR345dtb | data collection |
| PHASER | phasing |
| PHENIX | refinement |
| HKL-2000 | data reduction |
| HKL-2000 | data scaling |