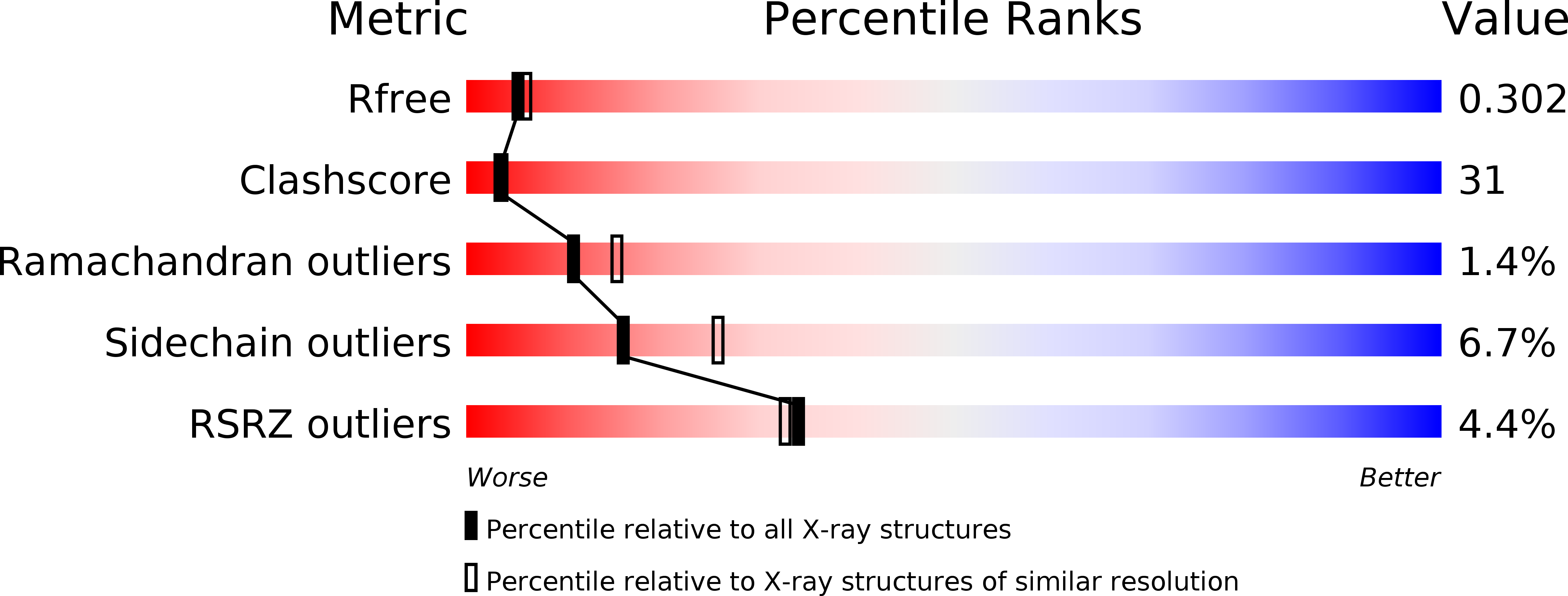
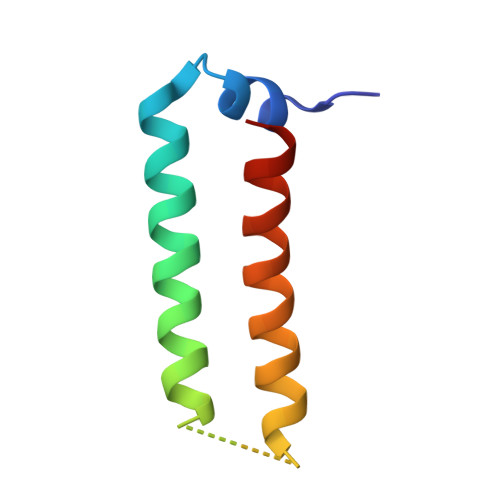
Crystal structure of the heteromolecular chaperone, AscE-AscG, from the type III secretion system in Aeromonas hydrophila
Chatterjee, C., Kumar, S., Chakraborty, S., Tan, Y.W., Leung, K.Y., Sivaraman, J., Mok, Y.K.(2011) PLoS One 6: e19208-e19208
- PubMed: 21559439
- DOI: https://doi.org/10.1371/journal.pone.0019208
- Primary Citation of Related Structures:
3PH0 - PubMed Abstract:
The putative needle complex subunit AscF forms a ternary complex with the chaperones AscE and AscG in the type III secretion system of Aeromonas hydrophila so as to avoid premature assembly. Previously, we demonstrated that the C-terminal region of AscG (residues 62-116) in the hetero-molecular chaperone, AscE-AscG, is disordered and susceptible to limited protease digestion.
Organizational Affiliation:
Department of Biological Sciences, National University of Singapore, Singapore.