Calmodulin bound to peptide from calmodulin kinase II (CaMKII)
Ng, H.L., Alber, T.A., Wand, A.J.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Calmodulin | 147 | Gallus gallus | Mutation(s): 0 Gene Names: CALM, CAM, RCJMB04_24e7 | 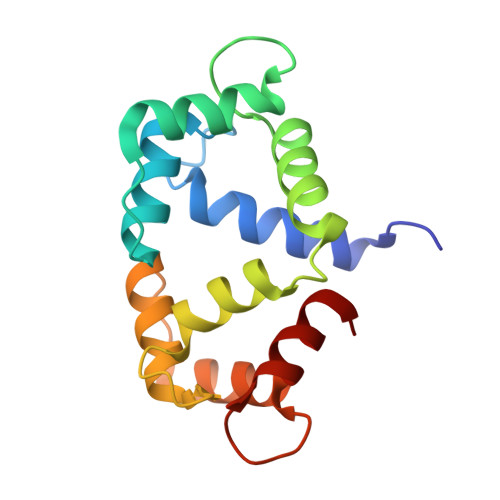 | |
UniProt | |||||
Find proteins for P62149 (Gallus gallus) Explore P62149 Go to UniProtKB: P62149 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P62149 | ||||
Sequence AnnotationsExpand | |||||
| |||||
Find similar proteins by: Sequence | 3D Structure
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Calcium/calmodulin-dependent protein kinase type II delta chain | 22 | Homo sapiens | Mutation(s): 0 EC: 2.7.11.17 |  | |
UniProt & NIH Common Fund Data Resources | |||||
Find proteins for Q13557 (Homo sapiens) Explore Q13557 Go to UniProtKB: Q13557 | |||||
PHAROS: Q13557 GTEx: ENSG00000145349 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q13557 | ||||
Sequence AnnotationsExpand | |||||
| |||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| CA Query on CA | C [auth A], D [auth A], E [auth A], F [auth A] | CALCIUM ION Ca BHPQYMZQTOCNFJ-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 69.599 | α = 90 |
| b = 38.409 | β = 125.27 |
| c = 75.012 | γ = 90 |
| Software Name | Purpose |
|---|---|
| Blu-Ice | data collection |
| EPMR | phasing |
| REFMAC | refinement |
| MOSFLM | data reduction |
| SCALA | data scaling |