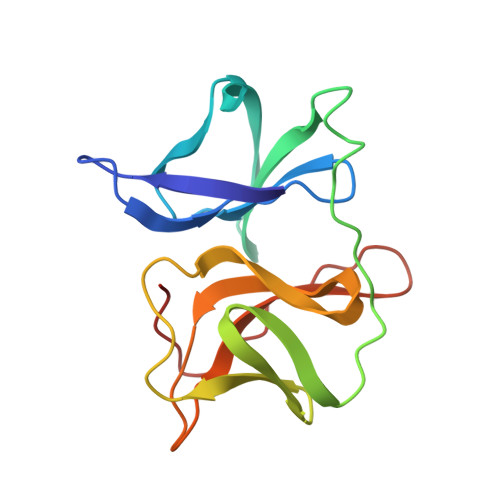
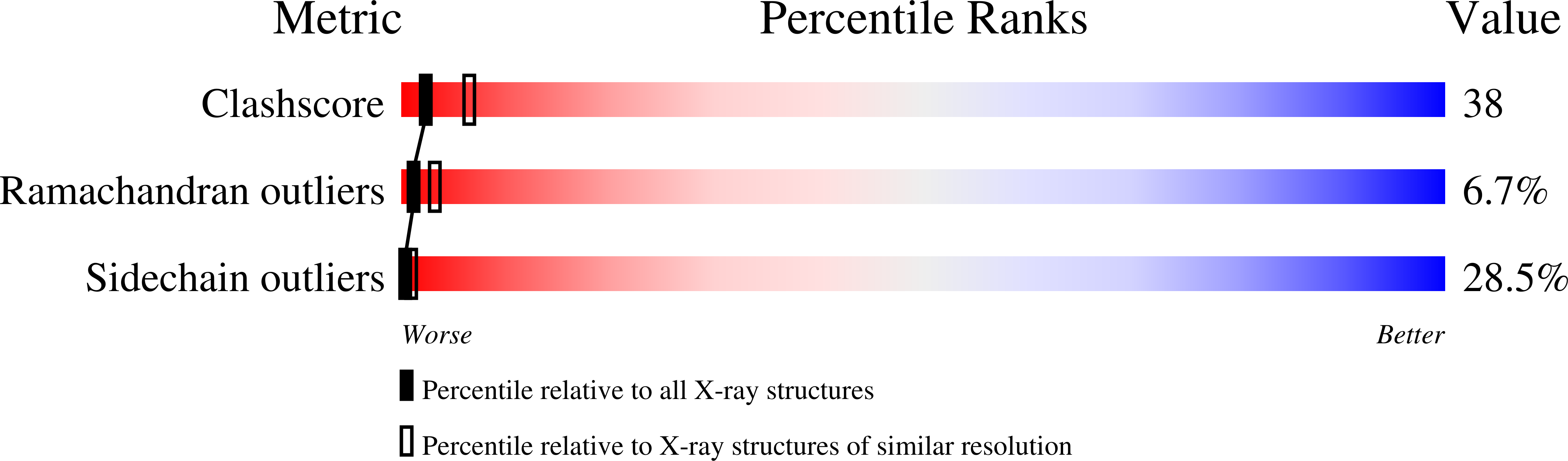Refined structure of Sindbis virus core protein and comparison with other chymotrypsin-like serine proteinase structures.
Tong, L., Wengler, G., Rossmann, M.G.(1993) J Mol Biol 230: 228-247
- PubMed: 8450538
- DOI: https://doi.org/10.1006/jmbi.1993.1139
- Primary Citation of Related Structures:
2SNV - PubMed Abstract:
Crystal forms 2 and 3 of Sindbis virus core protein have been refined to 2.8 A and 3.0 A resolution, respectively. The three independent molecular copies in the two crystal forms are essentially identical, except for regions where the molecules are involved in different crystal packing interactions. The overall polypeptide backbone fold of Sindbis virus core protein is similar to other chymotrypsin-like serine proteinase structures despite a lack of significant sequence homology. Detailed analysis revealed differences in the catalytic triad and the substrate binding pockets between the Sindbis virus core protein and the other serine proteinases. The catalytic aspartic acid residue (Asp163) and residue Asp214 (corresponding to Asp194 in chymotrypsin) are partially exposed to solvent in Sindbis virus core protein. Chymotrypsin Ser214, hydrogen bonded to the catalytic aspartic acid residue in all other serine proteinase structures, is changed to Leu231 in Sindbis virus core protein. Deletions in the loop regions on the surface of the protein account for the smaller size of the ordered part of Sindbis virus core protein (151 residues) as compared to chymotrypsin (236 residues), and permits the cis autocatalytic cleavage of the polyprotein to produce the viral capsid protein.
Organizational Affiliation:
Department of Biological Sciences, Purdue University West Lafayette, IN 47907.