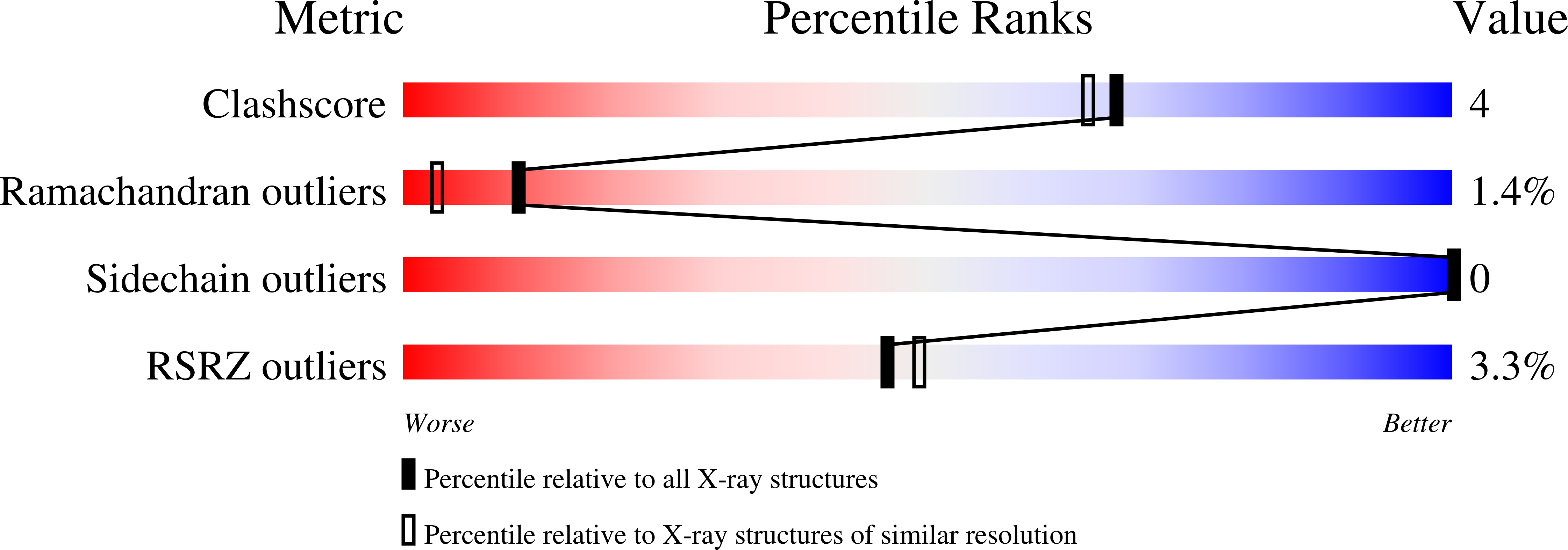Structural constraints on autoprocessing of the human nucleoporin Nup98.
Sun, Y., Guo, H.C.(2008) Protein Sci 17: 494-505
- PubMed: 18287282
- DOI: https://doi.org/10.1110/ps.073311808
- Primary Citation of Related Structures:
2Q5X, 2Q5Y - PubMed Abstract:
Nucleoporin Nup98, a 98-kDa protein component of the nuclear pore complex, plays an important role in both protein and RNA transport. During its maturation process, Nup98 undergoes post-translational autoproteolysis, which is critical for targeting to the NPC. Here we present high-resolution crystal structures of the C-terminal autoproteolytic domains of Nup98 (2.3 A for the wild type and 1.9 A for the S864A precursor), and propose a detailed autoproteolysis mechanism through an N-O acyl shift. Structural constraints are found at the autocleavage site, and could thus provide a driving force for autocleavage at the scissile peptide bond. Such structural constraints appear to be generated, at least in part, by anchoring a conserved phenylalanine side chain into a highly conserved hydrophobic pocket at the catalytic site. Our high-resolution crystal structures also reveal that three highly conserved residues, Tyr866, Gly867, and Leu868, provide most of the interactions between the autoproteolytic domain and the C-terminal tail. These results suggest that Nup98 may represent a new subtype of protein that utilizes autoprocessing to control biogenesis pathways and intracellular translocation.
Organizational Affiliation:
Department of Physiology and Biophysics, Boston University School of Medicine, Boston, Massachusetts 02118-2526, USA.