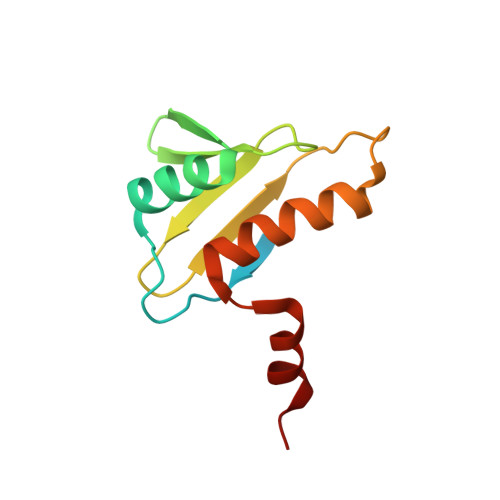
Solution structure of the hypothetical protein TA0095 from Thermoplasma acidophilum: A novel superfamily with a two-layer sandwich architecture
Leon, E., Yee, A., Ortiz, A.R., Santoro, J., Rico, M., Jimenez, M.A.(2007) Protein Sci 16: 2278-2286
- PubMed: 17766377
- DOI: https://doi.org/10.1110/ps.072869607
- Primary Citation of Related Structures:
2JOI - PubMed Abstract:
TA0095 is a 96-residue hypothetical protein from Thermoplasma acidophilum that exhibits no sequence similarity to any protein of known structure. Also, TA0095 is a member of the COG4004 orthologous group of unknown function found in Archaea bacteria. We determined its three-dimensional structure by NMR methods. The structure displays an alpha/beta two-layer sandwich architecture formed by three alpha-helices and five beta-strands following the order beta1-alpha1-beta2-beta3-beta4-beta5-alpha2-alpha3. Searches for structural homologs indicate that the TA0095 structure belongs to the TBP-like fold, constituting a novel superfamily characterized by an additional C-terminal helix. The TA0095 structure provides a fold common to the COG4004 proteins that will obviously belong to this new superfamily. Most hydrophobic residues conserved in the COG4004 proteins are buried in the structure determined herein, thus underlying their importance for structure stability. Considering that the TA0095 surface shows a large positively charged patch with a high degree of residue conservation within the COG4004 domain, the biological function of TA0095 and the rest of COG4004 proteins might occur through binding a negatively charged molecule. Like other TBP-like fold proteins, the COG4004 proteins might be DNA-binding proteins. The fact that TA0095 is shown to interact with large DNA fragments is in favor of this hypothesis, although nonspecific DNA binding cannot be ruled out.
Organizational Affiliation:
Instituto de Química Física Rocasolano, Madrid, Spain.