Crystal Structure of the Succinylglutamate Desuccinylase from Chromobacterium violaceum, Northeast Structural Genomics Target CvR22.
Forouhar, F., Abashidze, M., Conover, K., Acton, T.B., Montelione, G.T., Tong, L., Hunt, J.F.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Succinylglutamate desuccinylase | 341 | Chromobacterium violaceum ATCC 12472 | Mutation(s): 5 Gene Names: astE EC: 3.1 | 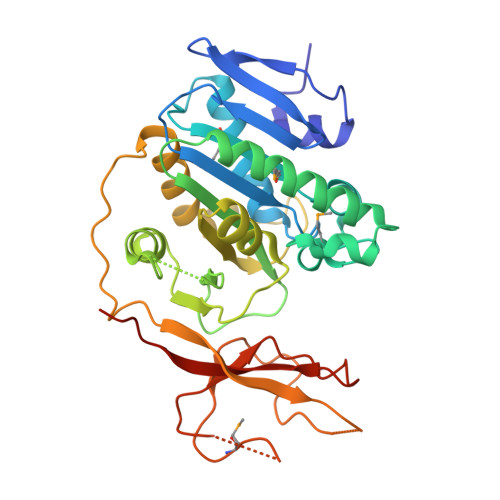 | |
UniProt | |||||
Find proteins for Q7NU26 (Chromobacterium violaceum (strain ATCC 12472 / DSM 30191 / JCM 1249 / NBRC 12614 / NCIMB 9131 / NCTC 9757)) Explore Q7NU26 Go to UniProtKB: Q7NU26 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q7NU26 | ||||
Sequence AnnotationsExpand | |||||
| |||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| ZN Query on ZN | C [auth A], D [auth B] | ZINC ION Zn PTFCDOFLOPIGGS-UHFFFAOYSA-N |  | ||
| Modified Residues 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| MSE Query on MSE | A, B | L-PEPTIDE LINKING | C5 H11 N O2 Se |  | MET |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 62.95 | α = 90 |
| b = 39.435 | β = 94.74 |
| c = 131.585 | γ = 90 |
| Software Name | Purpose |
|---|---|
| CNS | refinement |
| DENZO | data reduction |
| SCALEPACK | data scaling |
| SnB | phasing |
| SOLVE | phasing |
| RESOLVE | phasing |