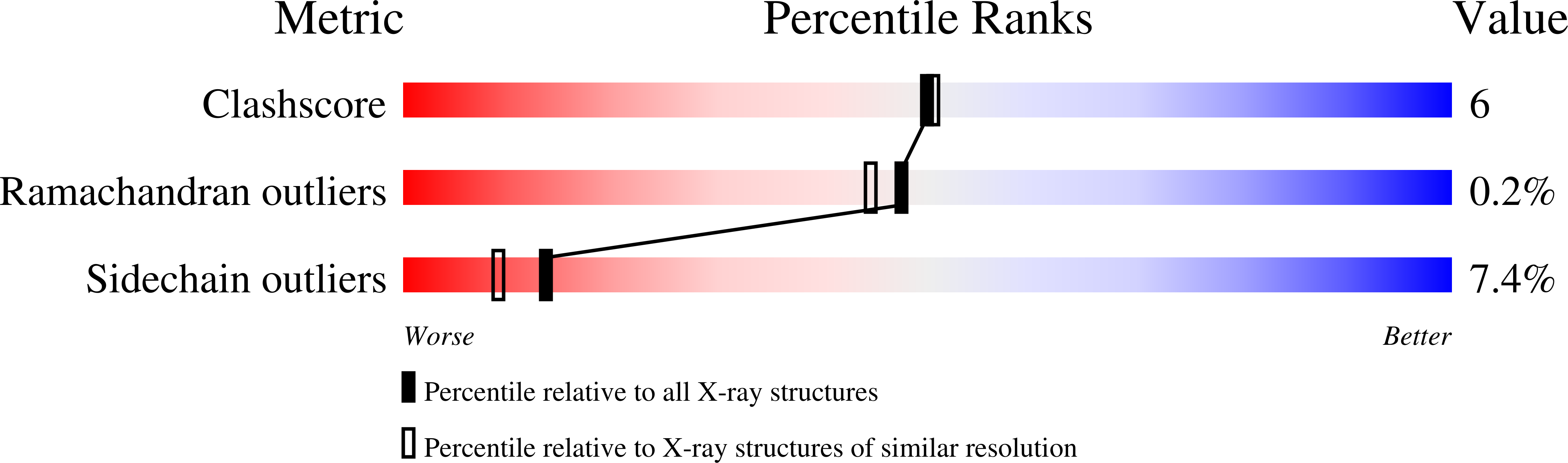Crystal structure of rabbit muscle glycogen phosphorylase a in complex with a potential hypoglycaemic drug at 2.0 A resolution
Oikonomakos, N.G., Chrysina, E.D., Kosmopoulou, M.N., Leonidas, D.D.(2003) Biochim Biophys Acta Proteins Proteom 1647: 325-332
- PubMed: 12686153
- DOI: https://doi.org/10.1016/s1570-9639(03)00085-2
- Primary Citation of Related Structures:
1LWN, 1LWO - PubMed Abstract:
CP320626 has been identified as a potent inhibitor, synergistic with glucose, of human liver glycogen phosphorylase a (LGPa), a possible target for type 2 diabetes therapy. CP320626 is also a potent inhibitor of human muscle GPa. In order to elucidate the structural basis of the mechanism of CP320626 inhibition, the structures of T state rabbit muscle GPa (MGPa) in complex with glucose and in complex with both glucose and CP320626 were determined at 2.0 A resolution, and refined to crystallographic R values of 0.179 (R(free)=0.218) and 0.207 (R(free)=0.235), respectively. CP320626 binds at the new allosteric site, some 33 A from the catalytic site, where glucose binds. The binding of CP320626 to MGPa does not promote extensive conformational changes except for small shifts of the side chain atoms of residues R60, V64, and K191. Both CP320626 and glucose promote the less active T state, while structural comparisons of MGPa-glucose-CP320626 complex with LGPa complexed with a related compound (CP403700) and a glucose analogue inhibitor indicate that the residues of the new allosteric site, conserved in the two isozymes, show no significant differences in their positions.
Organizational Affiliation:
Institute of Biological Research and Biotechnology, The National Hellenic Research Foundation, 48 Vas. Constantinou Avenue, Athens 11635, Greece. ngo@eie.gr