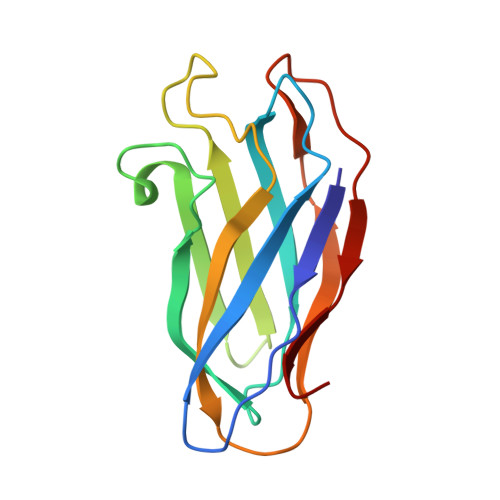
A cohesin domain from Clostridium thermocellum: the crystal structure provides new insights into cellulosome assembly.
Shimon, L.J., Bayer, E.A., Morag, E., Lamed, R., Yaron, S., Shoham, Y., Frolow, F.(1997) Structure 5: 381-390
- PubMed: 9083107
- DOI: https://doi.org/10.1016/s0969-2126(97)00195-0
- Primary Citation of Related Structures:
1ANU - PubMed Abstract:
The scaffoldin component of the cellulolytic bacterium Clostridium thermocellum is a non-hydrolytic protein which organizes the hydrolytic enzymes in a large complex, called the cellulosome. Scaffoldin comprises a series of functional domains, amongst which is a single cellulose-binding domain and nine cohesin domains which are responsible for integrating the individual enzymatic subunits into the complex. The cohesin domains are highly conserved in their primary amino acid sequences. These domains interact with a complementary domain, termed the dockerin domain, one of which is located on each enzymatic subunit. The cohesin-dockerin interaction is the crucial interaction for complex formation in the cellulosome. The determination of structural information about the cohesin domain will provide insights into cellulosome assembly and activity.
Organizational Affiliation:
Faculty of Chemistry, The Weizmann Institute of Science, Rehovot 76100, Israel. cvlinda@weizmann.weizmann.ac.il