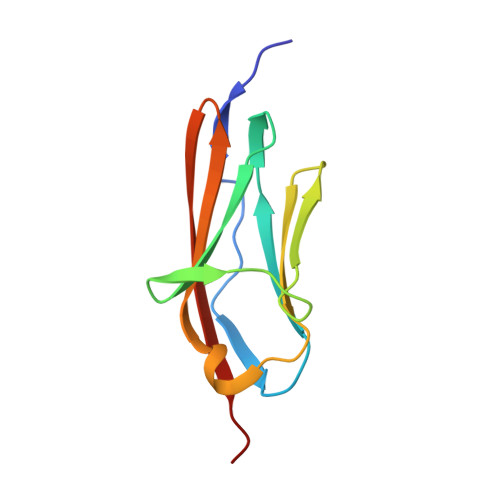
Solution structure of the first immunoglobulin domain of human myotilin
Heikkinen, O., Permi, P., Koskela, H., Carpen, O., Ylanne, J., Kilpelainen, I.(2009) J Biomol NMR 44: 107-112
- PubMed: 19418025
- DOI: https://doi.org/10.1007/s10858-009-9320-4
- Primary Citation of Related Structures:
2KDG - PubMed Abstract:
Myotilin is a 57 kDa actin-binding and -bundling protein that consists of a unique serine-rich amino-terminus, two Ig-domains and a short carboxy-terminus with a PDZ-binding motif. Myotilin localizes in sarcomeric Z-discs, where it interacts with several sarcomeric proteins. Point mutations in myotilin cause muscle disorders morphologically highlighted by sarcomeric disarray and aggregation. The actin-binding and dimerization propensity of myotilin has been mapped to the Ig-domains. Here we present high-resolution structure of the first Ig-domain of myotilin (MyoIg1) determined with solution state NMR spectroscopy. Nearly complete chemical shift assignments of MyoIg1 were achieved despite several missing backbone 1H-15N-HSQC signals. The structure derived from distance and dihedral angle restraints using torsion angle dynamics was further refined using molecular dynamics. The structure of MyoIg1 exhibits I-type Ig-fold. The absence of several backbone 1H-15N-HSQC signals can be explained by conformational exchange taking place at the hydrophobic core of the protein.
Organizational Affiliation:
Laboratory of Organic Chemistry, Department of Chemistry, University of Helsinki, P.O. Box 55, 00014 Helsinki, Finland. Outi.K.Heikkinen@helsinki.fi