Structural and dynamic insights into the biased signaling mechanism of the human kappa opioid receptor.
Suno-Ikeda, C., Nishikawa, R., Suzuki, R., Yokoi, S., Iwata, S., Takai, T., Ogura, T., Hirose, M., Tokuda, A., Katamoto, R., Inoue, A., Asai, E., Kise, R., Sugita, Y., Kato, T., Nagase, H., Mitsutake, A., Saitoh, T., Katayama, K., Inoue, A., Kandori, H., Kobayashi, T., Suno, R.(2025) Nat Commun 16: 9392-9392
- PubMed: 41152269 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41467-025-64882-1
- Primary Citation Related Structures:
9V6O, 9W49 - PubMed Abstract:
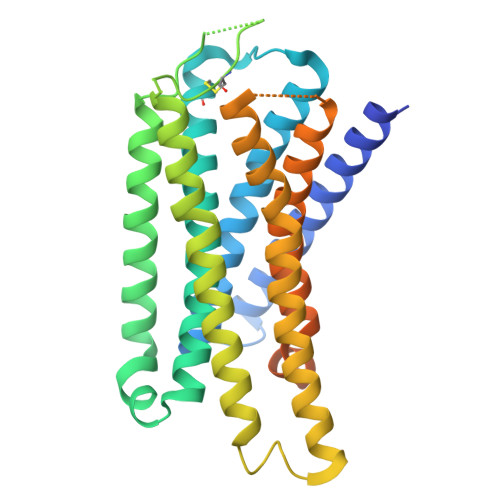
The κ-opioid receptor (KOR) is a member of the G protein-coupled receptor (GPCR) family, modulating cellular responses through transducers such as G proteins and β-arrestins. G-protein-biased KOR agonists aim to retain analgesic and antipruritic actions while limiting aversion and sedation. Aiming to inform G-biased KOR agonist design, we analyze signaling-relevant residues from structural and dynamic views. Here we show, using multiple complementary methods, shared residues that determine β-arrestin recruitment by nalfurafine and U-50,488H. Cryo-electron microscopy structures of the KOR-G i signaling complexes identify the ligand binding mode in the activated state. Vibrational spectroscopy reveals ligand-induced conformational changes. Cell-based mutant experiments pinpoint four amino acids (K227 5.40 , C286 6.47 , H291 6.52 , and Y312 7.34 ; Ballesteros-Weinstein numbering is shown in superscript) that play crucial roles in β-arrestin recruitment. Furthermore, MD simulations revealed that the four mutants tend to adopt conformations with reduced β-arrestin recruitment activity. Our research findings provide a foundation for enhancing KOR-mediated therapeutic effects while minimizing unwanted side effects by targeting specific residues within the KOR ligand-binding pocket, including K227 5.40 and Y312 7.34 , which have previously been implicated in biased signaling.
- Department of Medical Chemistry, Kansai Medical University, Hirakata, Japan.
Organizational Affiliation: