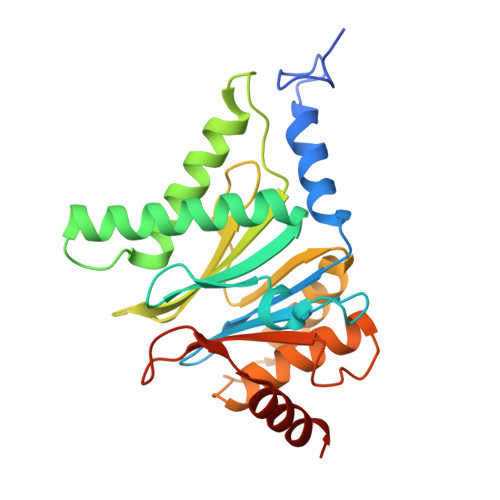
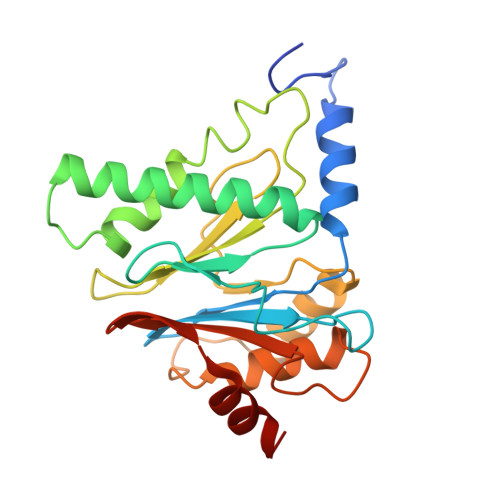
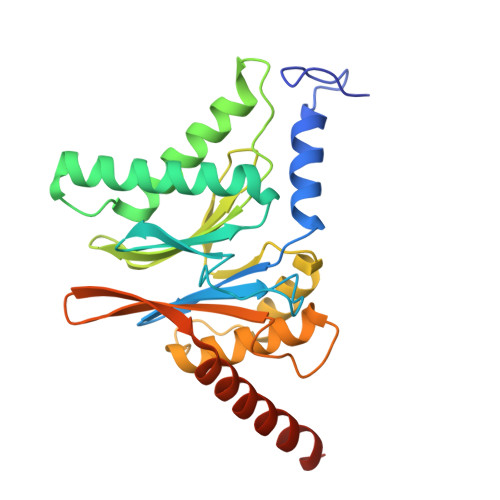
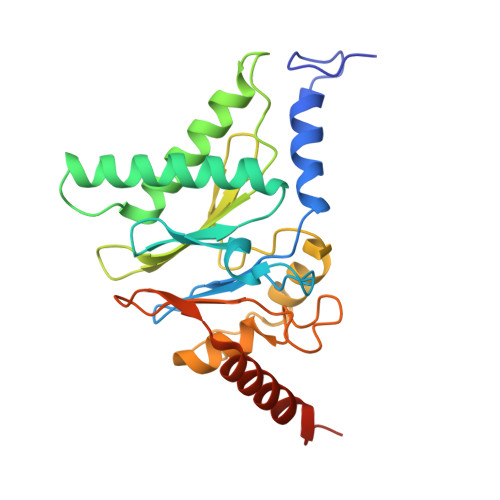
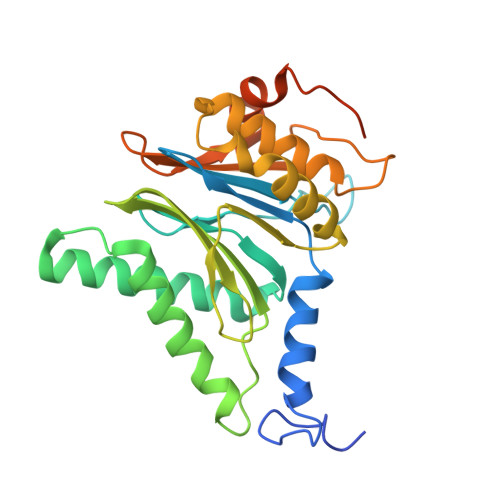
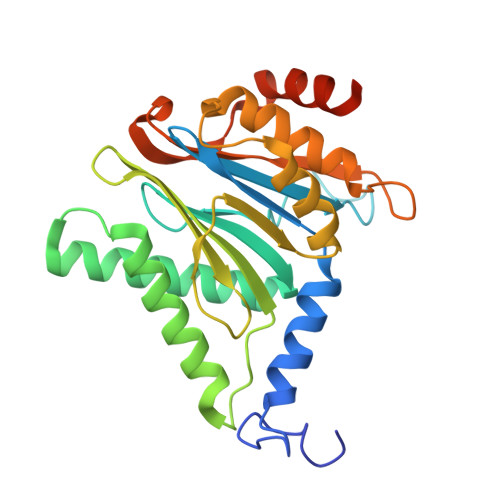
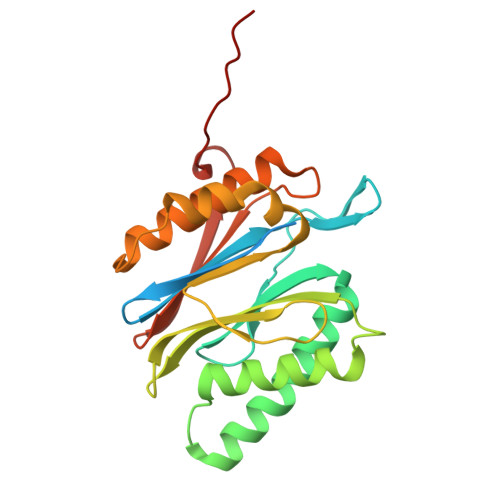
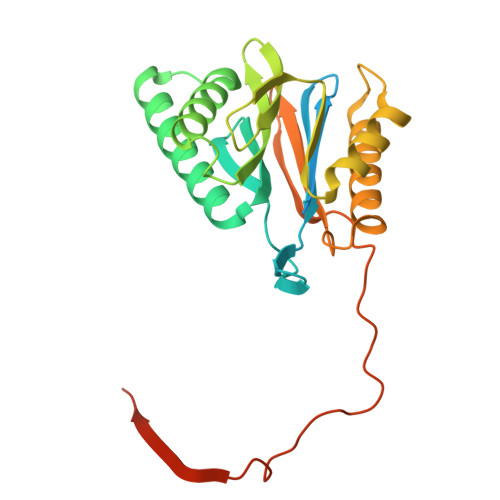
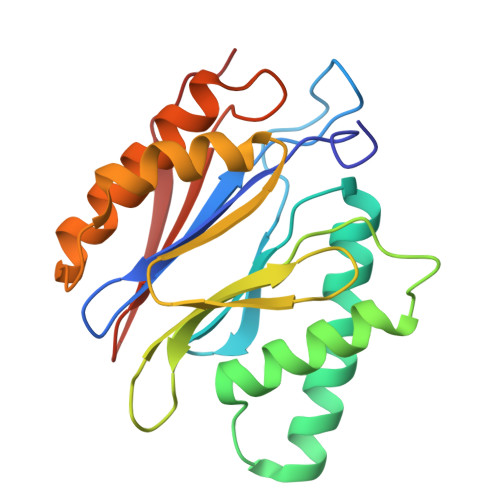
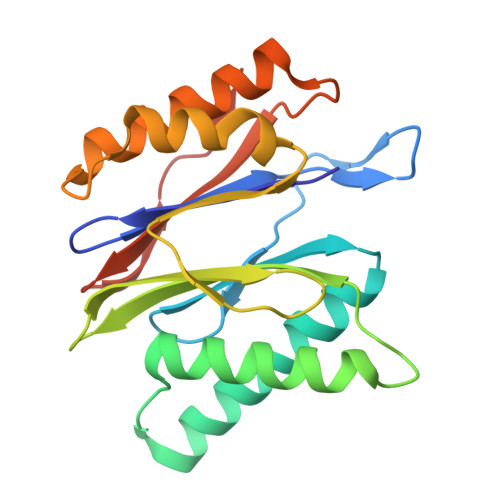
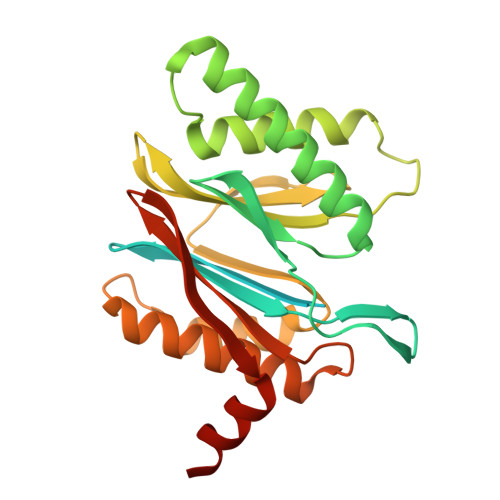
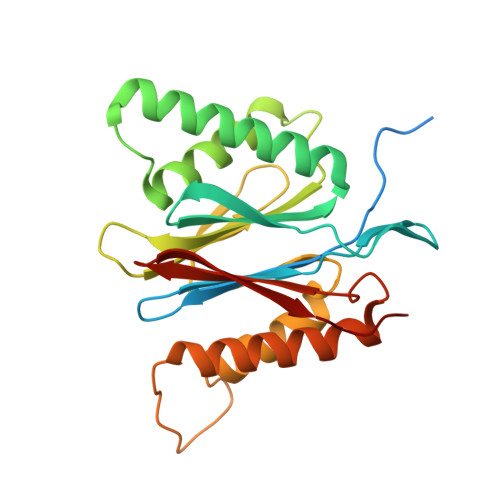
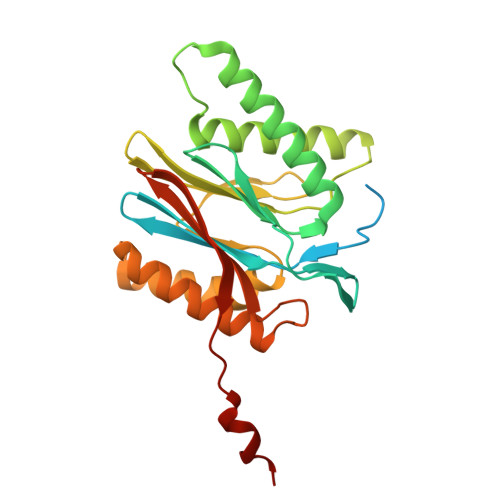
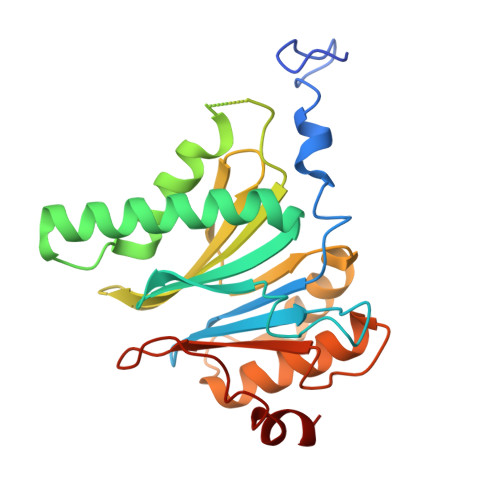
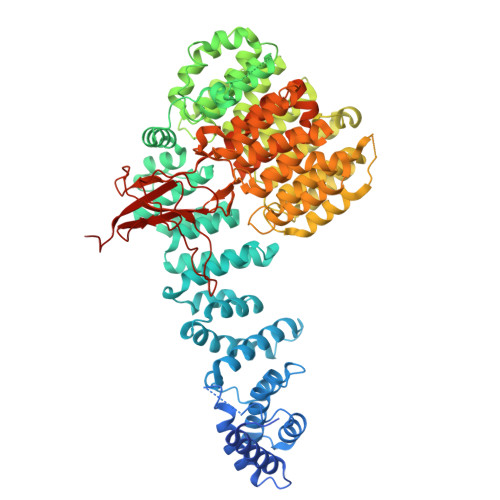
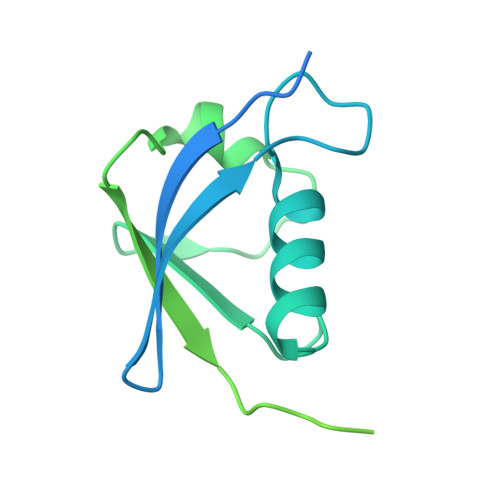
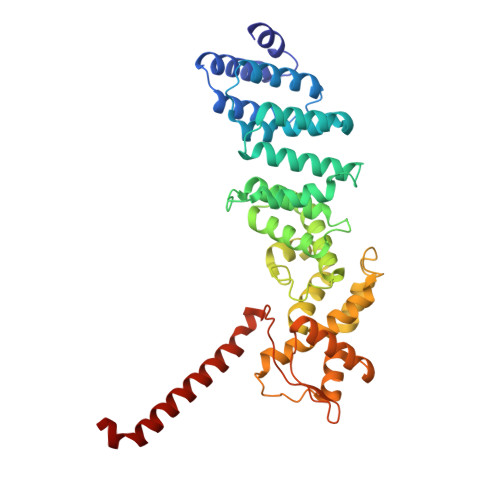
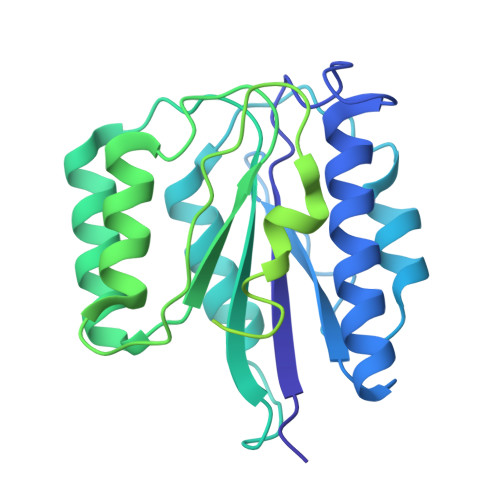
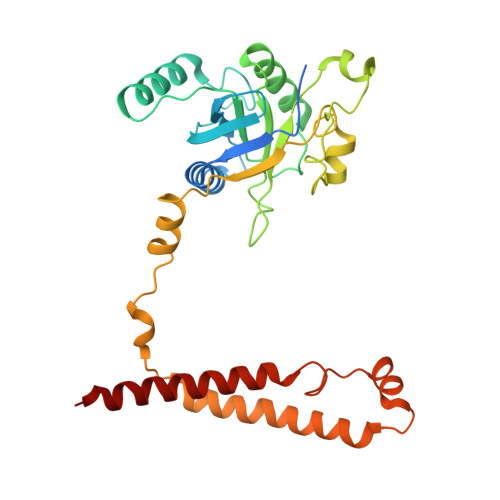
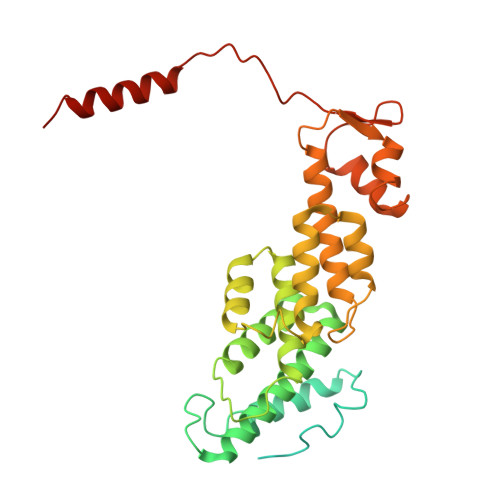
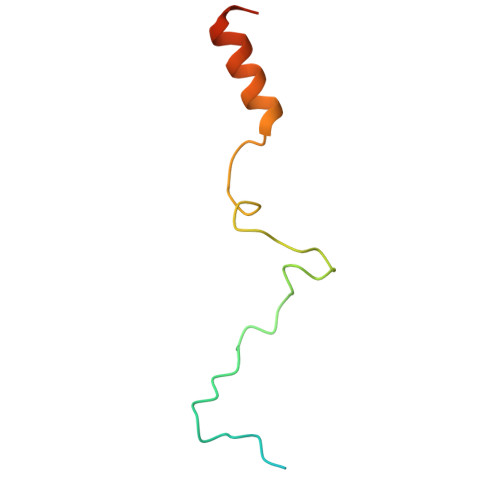
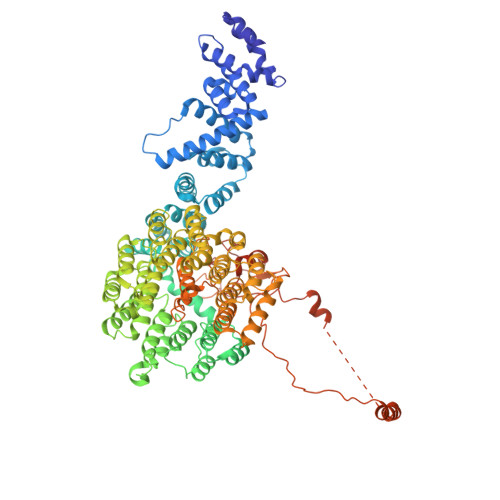
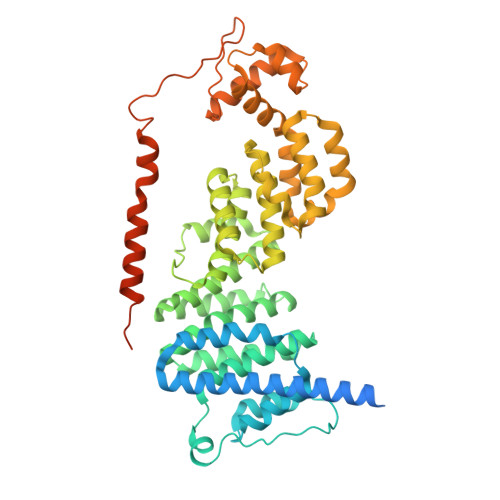
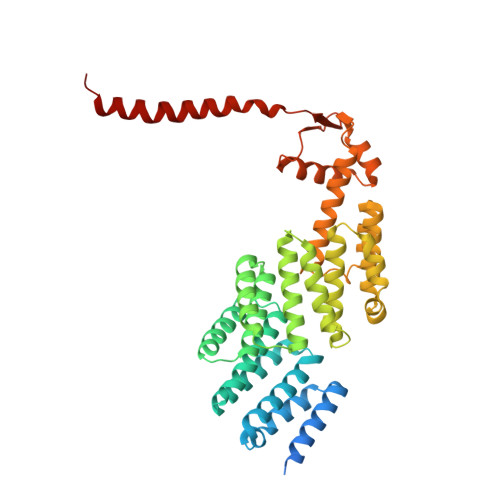
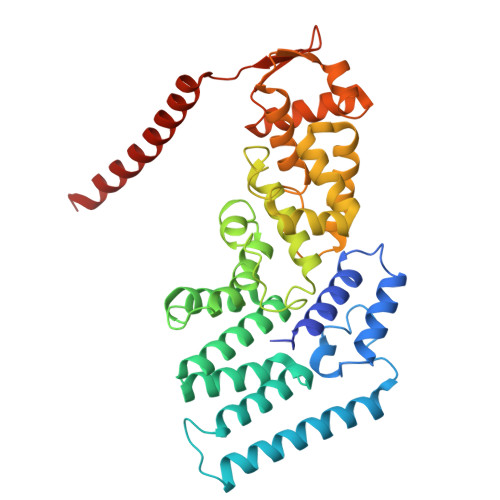
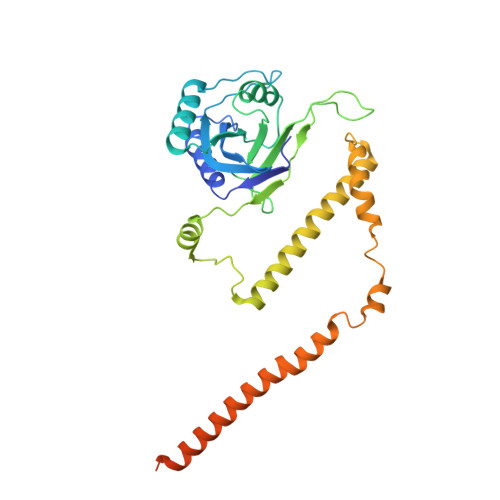
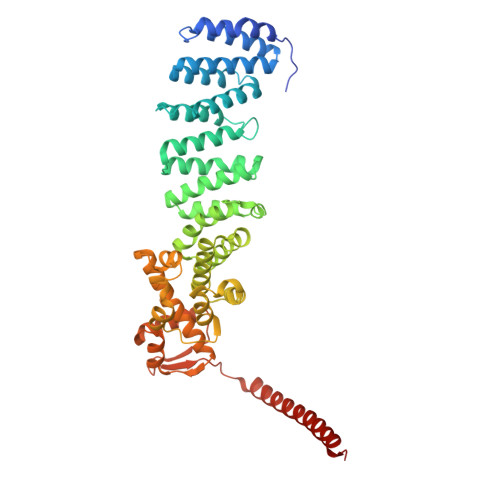
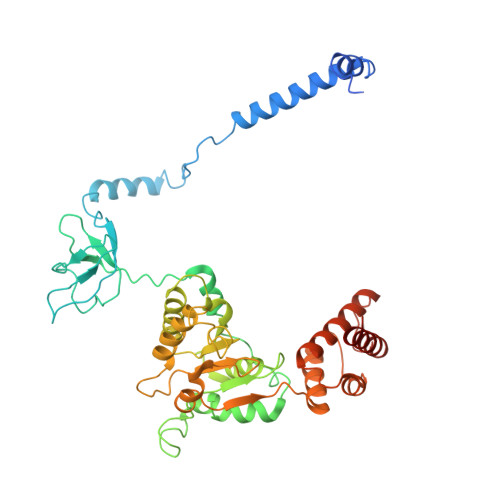
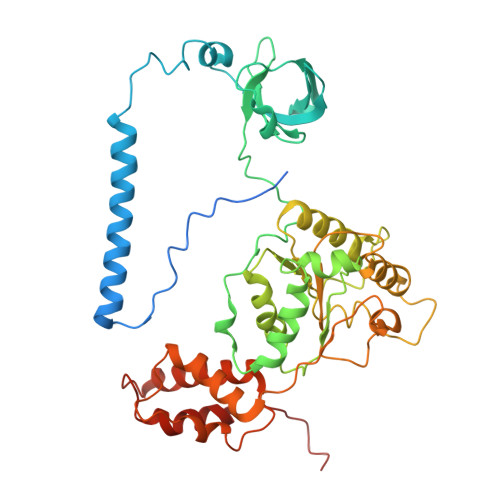
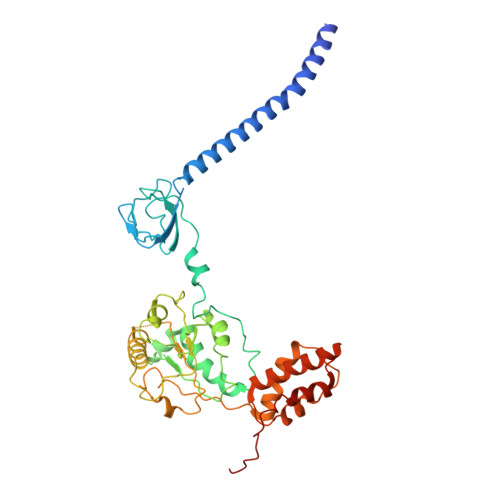
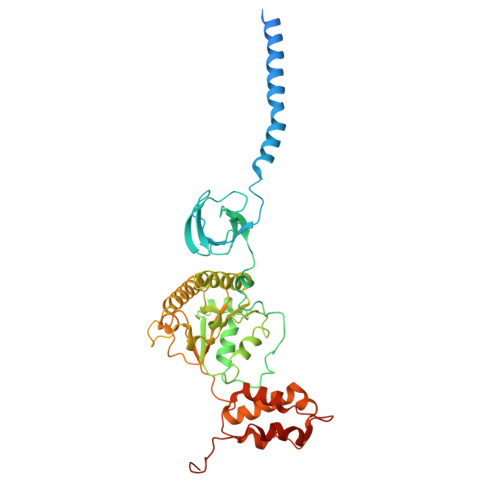
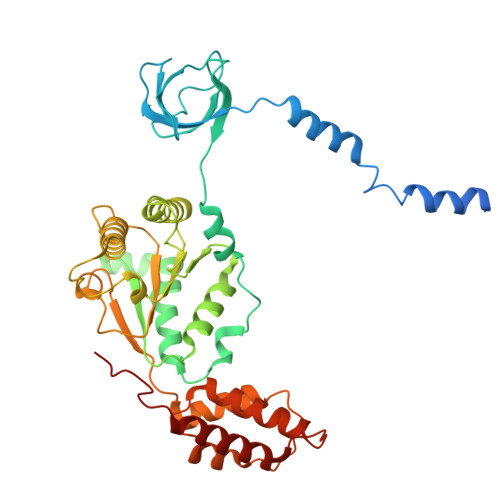
Structures of the 26 S proteasome in complex with the Hsp70 co-chaperone Bag1 reveal a mechanism for direct substrate transfer.
Maestro-Lopez, M., Cheng, T.C., Muntaner, J., Menendez, M., Alonso, M., Schweitzer, A., Ishizaka, M., Tomko Jr., R.J., Cuellar, J., Valpuesta, J.M., Sakata, E.(2026) Sci Adv 12: eadz3026-eadz3026
- PubMed: 41719407 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1126/sciadv.adz3026
- Primary Citation Related Structures:
9HEU, 9SZT, 9SZV - PubMed Abstract:
Coupling between the chaperone and degradation systems, particularly under stress, is essential for eliminating unfolded proteins. The co-chaperone Bag1 links Hsp70 to the 26 S proteasome, recruiting Hsp70-bound clients for proteasomal degradation. Here, we present cryo-electron microscopy structures of the Bag1-bound 26 S proteasome, revealing unprecedented conformational rearrangements within the 19 S regulatory particle. Bag1 binding to the Rpn1 induces a marked reconfiguration of AAA + adenosine triphosphatase (ATPase) ring, disrupting its canonical spiral staircase and remodeling the central channel architecture. This reconfiguration generates a large cavity above the substrate entry gate of the 20 S core particle. The conserved pore-2 loops of ATPases Rpt2 and Rpt5 play critical roles in opening of the 20 S gate, enabling substrate entry into proteolytic chamber independently of ubiquitination. These findings suggest a previously unknown mechanism of the proteasomal degradation, by which remodeling the central cavity and 20 S gate in the presence of Bag1, possibly bypassing the need for ubiquitination.
- Centro Nacional de Biotecnología (CNB-CSIC), 28049 Madrid, Spain.
Organizational Affiliation: