Cryo-EM structure of TCRpriv/pMHC
Akil, C., Zhu, Y., Hardenbrook, N., Zhang, P.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| T cell receptor alpha chain | 205 | Homo sapiens | Mutation(s): 0 | 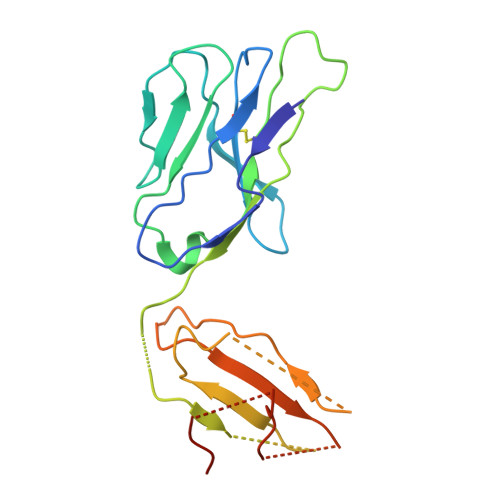 | |
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| T cell receptor beta chain | 242 | Homo sapiens | Mutation(s): 0 | 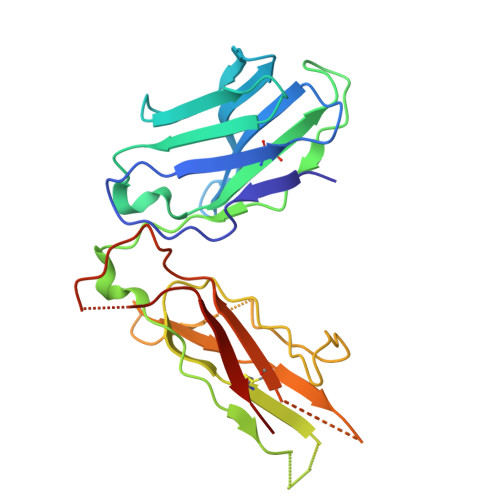 | |
Entity ID: 3 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| SARS-CoV-2 ORF3a_207-215 epitope | C [auth D] | 9 | Homo sapiens | Mutation(s): 0 | 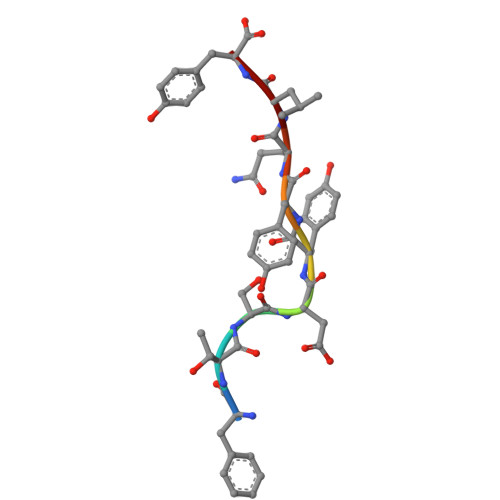 |
Entity ID: 4 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| MHC class I antigen | D [auth E] | 272 | Homo sapiens | Mutation(s): 0 Gene Names: HLA-A | 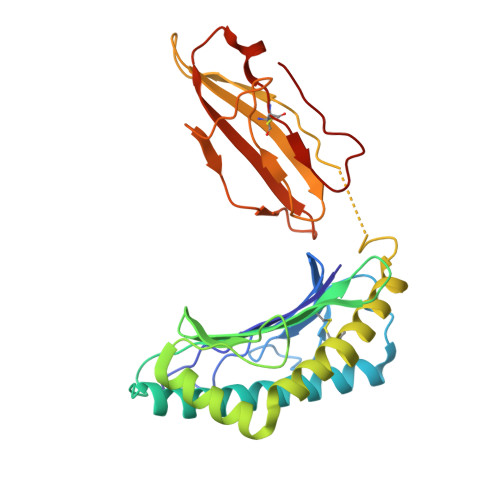 |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A7WPI8 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 5 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Beta-2-microglobulin | E [auth F] | 99 | Homo sapiens | Mutation(s): 0 Gene Names: B2M, CDABP0092, HDCMA22P |  |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P61769 GTEx: ENSG00000166710 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P61769 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Task | Software Package | Version |
|---|---|---|
| MODEL REFINEMENT | PHENIX | |
| RECONSTRUCTION | RELION | 5 |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Chinese Academy of Sciences | China | 2024-I2M-2-001-1 |
| Wellcome Trust | United Kingdom | 203141/Z/16/Z |