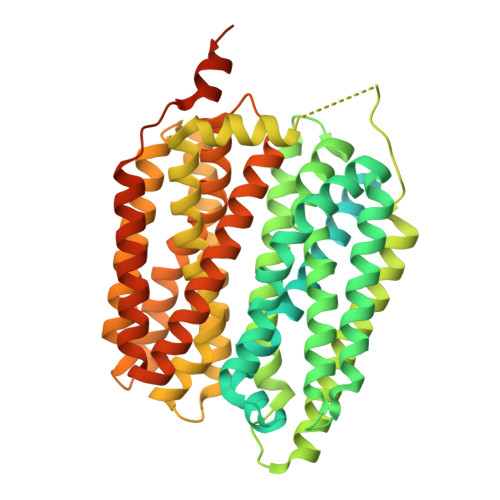
Structural and biochemical comparison of the FLVCR and CTL membrane protein families in eukaryotes.
Nel, L., Driller, J.H., Driller, R., Frain, K.M., Pedersen, B.P.(2026) Life Sci Alliance 9
- PubMed: 42114998 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.26508/lsa.202503583
- Primary Citation Related Structures:
9F63, 9QU3, 9QU4 - PubMed Abstract:
The organic cation choline is essential for eukaryotic metabolism. Recently, the feline leukemia virus subgroup C receptor-related (FLVCR, SLC49) family was demonstrated as central for basal choline transport, questioning the role of the choline transporter-like (CTL, SLC44) family in this capacity. Here, we use Xenopus laevis oocytes to confirm that FLVCR1 (SLC49A1) and FLVCR2 (SLC49A2) proteins are choline transporters. CTL1 (SLC44A1) does not transport choline under the same conditions, supported by other CTL proteins, Arabidopsis thaliana CherI and Saccharomyces cerevisiae PNS1, which also display no choline transport activity. We present the atomic structures of FLVCR2, CTL1, and PNS1. The 3.4 Å cryo-EM structure of FLVCR2 has choline in the binding pocket. The 3.3 Å cryo-EM structure of CTL1 and the 2.7 Å crystal structure of PNS1 reveal an unusual protein fold, weakly related to the mitochondrial carrier family (SLC25). The unusual fold appears incompatible with transmembrane transport and implies a different and, so far, unknown function for CTL proteins. Our results support FLVCR proteins as choline transporters and suggest a nontransport role for CTL proteins.
- Department of Molecular Biology and Genetics, Aarhus University, Aarhus, Denmark.
Organizational Affiliation: