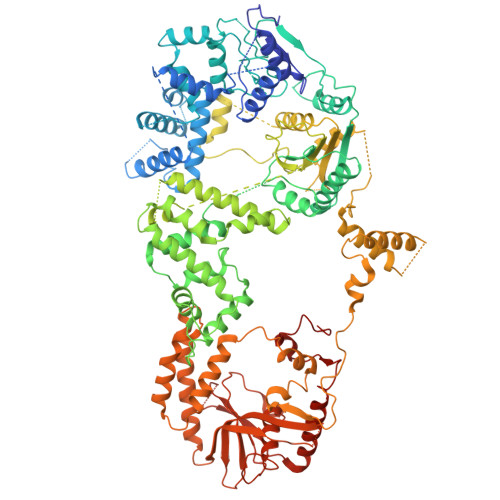
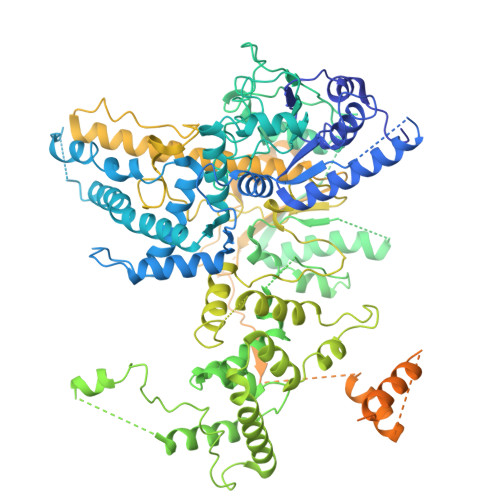
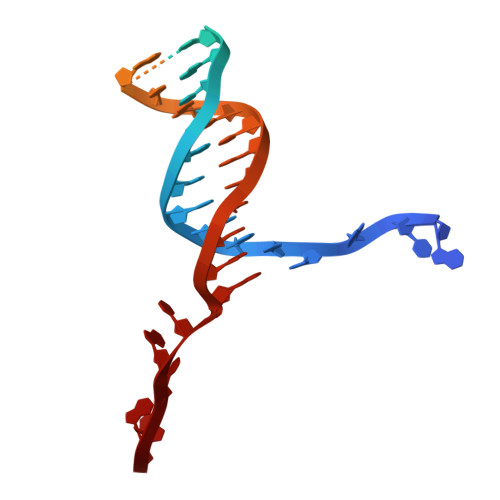
Structure of an initiation complex of mycobacterial helicase-nuclease AdnAB at a blunt double-strand break reveals local melting that engages the 3' tracking strand at the ratchet pawl of the helicase motor.
Warren, G.M., Jason de la Cruz, M., Goldgur, Y., Shuman, S.(2026) Nucleic Acids Res 54
- PubMed: 41854076 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1093/nar/gkag243
- Primary Citation Related Structures:
9NP6 - PubMed Abstract:
Mycobacterial AdnAB is a heterodimeric helicase-nuclease that initiates homologous recombination by resecting double-strand breaks. The AdnB subunit hydrolyzes ATP to drive single-nucleotide steps of 3'-to-5' translocation of AdnAB on the tracking DNA strand via a ratchet-like mechanism. AdnB Trp325, which makes a π stack on a nucleobase 5' of a flipped-out nucleoside, is the ratchet pawl without which ATP hydrolysis is mechanically futile. Here we report a cryo-EM (cryogenic electron microscopy) structure of AdnAB in complex with a blunt-ended DNA. AdnAB forms an initiation complex by melting five terminal base pairs and threading the splayed out 5' and 3' single strands into the AdnA nuclease domain and the AdnB motor domain, respectively. The melted 5-nucleotide 3' tracking strand is in-gear with respect to the ratchet and pawl of the AdnB motor. An analogous π-stacking aromatic pawl is present in the motor subunit of the bacterial end-resection enzymes Bacillus AddAB and Escherichia coli RecBCD. Our results highlight a common theme whereby binding of the motor-nuclease to a blunt DSB end, in the absence of ATP hydrolysis, is coupled to local melting of the terminal base pairs that suffices to engage the "clutch" of the motor on the tracking strand.
- Molecular Biology Program, Memorial Sloan Kettering Cancer Center, NY, NY 10065, United States.
Organizational Affiliation: