Crystal structure and proposed mechanism of an DUF3237 protein from Aspergillus.
Li, C.P., Fu, Y.Z., Jiang, Q.Y., Zhu, L.J., Gu, L., Zhao, Y.C., Yang, M.H., Kong, L.Y.To be published.
Experimental Data Snapshot
Starting Model: in silico
View more details
wwPDB Validation 3D Report Full Report
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| DUF3237 protein | 157 | Aspergillus | Mutation(s): 0 | 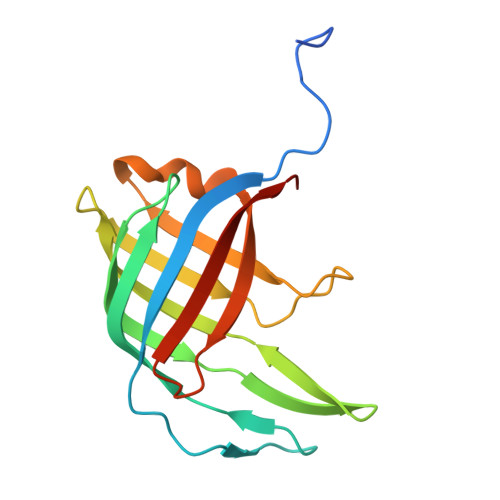 | |
UniProt | |||||
Find proteins for A0A1L9TCT0 (Aspergillus sydowii CBS 593.65) Explore A0A1L9TCT0 Go to UniProtKB: A0A1L9TCT0 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A0A1L9TCT0 | ||||
Sequence AnnotationsExpand | |||||
| |||||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 45.1 | α = 90 |
| b = 70.52 | β = 90 |
| c = 88.85 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| Aimless | data scaling |
| XDS | data reduction |
| PHENIX | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Ministry of Education (MoE, China) | China | B18056 |