Neddylation induces an intermediate auto-inhibited CRL2FEM1B E3 ubiquitin ligase
Dai, Z., Liang, L., Yin, Y.X.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Cullin-2 | A, B [auth D] | 751 | Homo sapiens | Mutation(s): 0 Gene Names: CUL2 | 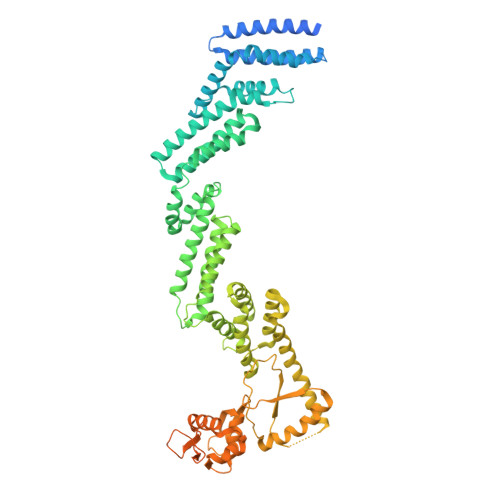 |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: Q13617 GTEx: ENSG00000108094 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q13617 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| E3 ubiquitin-protein ligase RBX1 | C [auth E], F [auth H] | 108 | Homo sapiens | Mutation(s): 0 Gene Names: RBX1, RNF75, ROC1 EC: 2.3.2.27 (PDB Primary Data), 2.3.2.32 (PDB Primary Data) | 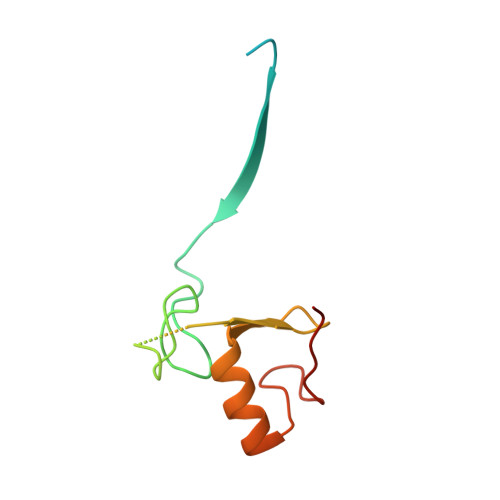 |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P62877 GTEx: ENSG00000100387 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P62877 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 3 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Elongin-C | D [auth F] | 98 | Homo sapiens | Mutation(s): 0 Gene Names: ELOC, TCEB1 | 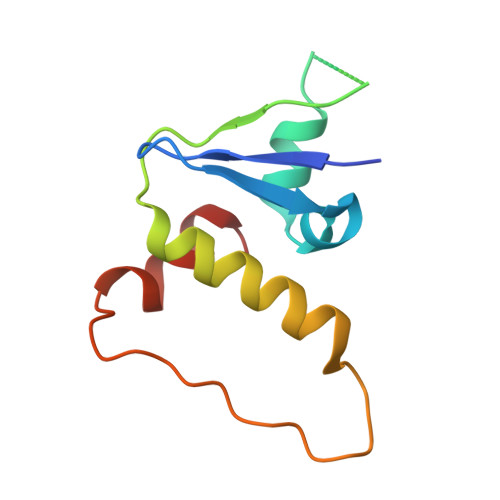 |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: Q15369 GTEx: ENSG00000154582 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q15369 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 4 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Elongin-B | E [auth G] | 104 | Homo sapiens | Mutation(s): 0 Gene Names: ELOB, TCEB2 | 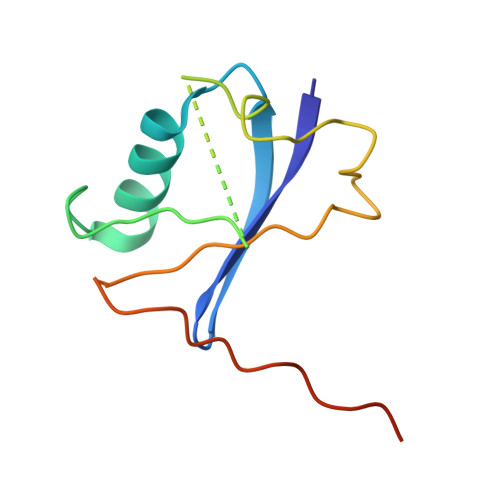 |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: Q15370 GTEx: ENSG00000103363 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q15370 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 5 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Protein fem-1 homolog B | G [auth I] | 628 | Homo sapiens | Mutation(s): 0 Gene Names: FEM1B, F1AA, KIAA0396 | 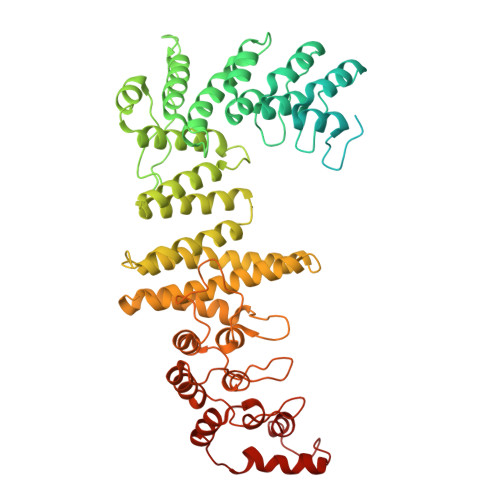 |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: Q9UK73 GTEx: ENSG00000169018 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q9UK73 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 6 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Maltose/maltodextrin-binding periplasmic protein,Folliculin interacting protein 1 | H [auth J] | 467 | Escherichia coli K-12, Homo sapiens This entity is chimeric | Mutation(s): 0 Gene Names: malE, b4034, JW3994, FNIP1 | 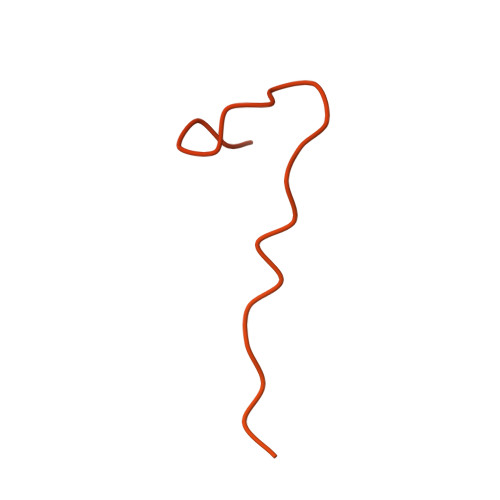 |
UniProt & NIH Common Fund Data Resources | |||||
GTEx: ENSG00000217128 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | J3KNG8 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| ZN (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | I [auth E] J [auth E] K [auth E] L [auth H] M [auth H] | ZINC ION Zn PTFCDOFLOPIGGS-UHFFFAOYSA-N |  | ||
| Task | Software Package | Version |
|---|---|---|
| MODEL REFINEMENT | PHENIX | |
| RECONSTRUCTION | cryoSPARC |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Natural Science Foundation of China (NSFC) | China | 81621063, 31800626 |