Molecular mechanism of transition-state inhibitors of bacterial antibiotic efflux pumps
Boernsen, C., Mueller, R.T., Pos, K.M.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Multidrug efflux pump subunit AcrB | 1,057 | Escherichia coli K-12 | Mutation(s): 2 Gene Names: acrB, acrE, b0462, JW0451 | 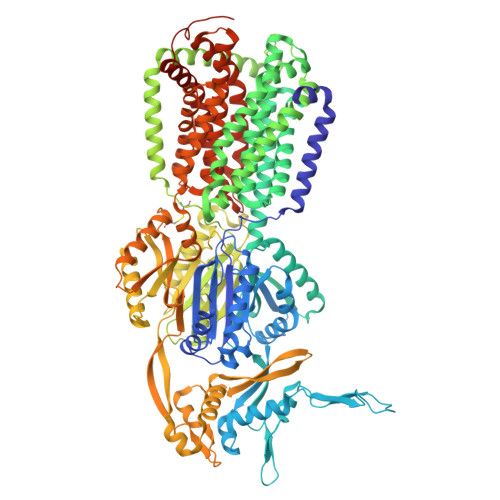 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P31224 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| DARPIN | 169 | synthetic construct | Mutation(s): 0 | 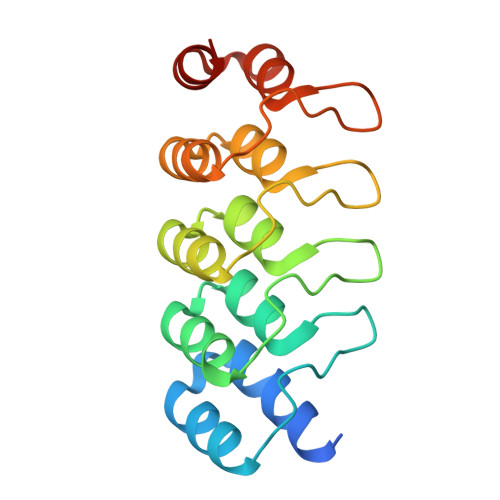 | |
| Ligands 9 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| LMT Download:Ideal Coordinates CCD File | BA [auth B] F [auth A] G [auth A] GA [auth C] H [auth A] | DODECYL-BETA-D-MALTOSIDE C24 H46 O11 NLEBIOOXCVAHBD-QKMCSOCLSA-N |  | ||
| C14 Download:Ideal Coordinates CCD File | EA [auth C] | TETRADECANE C14 H30 BGHCVCJVXZWKCC-UHFFFAOYSA-N |  | ||
| D12 Download:Ideal Coordinates CCD File | IA [auth C], L [auth A], LA [auth C], Y [auth B] | DODECANE C12 H26 SNRUBQQJIBEYMU-UHFFFAOYSA-N |  | ||
| D10 Download:Ideal Coordinates CCD File | I [auth A] KA [auth C] MA [auth C] Q [auth B] S [auth B] | DECANE C10 H22 DIOQZVSQGTUSAI-UHFFFAOYSA-N |  | ||
| DD9 Download:Ideal Coordinates CCD File | OA [auth C] | nonane C9 H20 BKIMMITUMNQMOS-UHFFFAOYSA-N |  | ||
| OCT Download:Ideal Coordinates CCD File | HA [auth C], JA [auth C] | N-OCTANE C8 H18 TVMXDCGIABBOFY-UHFFFAOYSA-N |  | ||
| SO4 Download:Ideal Coordinates CCD File | SA [auth C] | SULFATE ION O4 S QAOWNCQODCNURD-UHFFFAOYSA-L |  | ||
| GOL Download:Ideal Coordinates CCD File | CA [auth B] DA [auth B] FA [auth C] K [auth A] M [auth A] | GLYCEROL C3 H8 O3 PEDCQBHIVMGVHV-UHFFFAOYSA-N |  | ||
| HEX Download:Ideal Coordinates CCD File | AA [auth B], NA [auth C], PA [auth C] | HEXANE C6 H14 VLKZOEOYAKHREP-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 145.71 | α = 90 |
| b = 165.3 | β = 90 |
| c = 244.89 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PDB-REDO | refinement |
| XDS | data reduction |
| XDS | data scaling |
| PHASER | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Swiss National Science Foundation | Switzerland | -- |
| University of Zurich | Switzerland | -- |
| German Research Foundation (DFG) | Germany | SFB 807 |