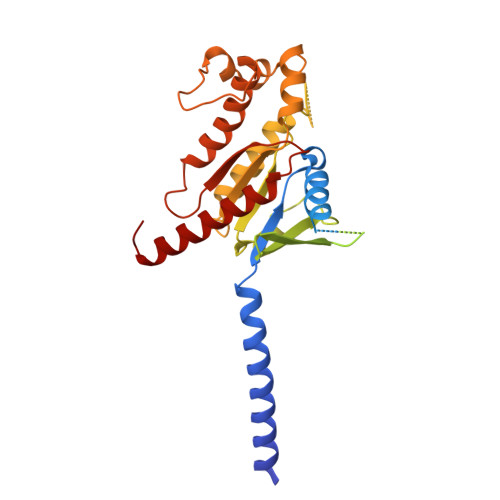
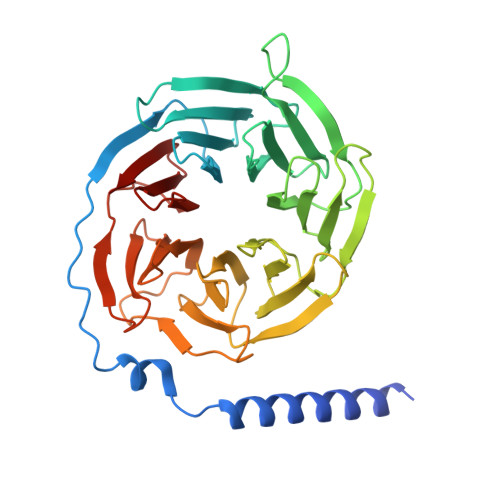
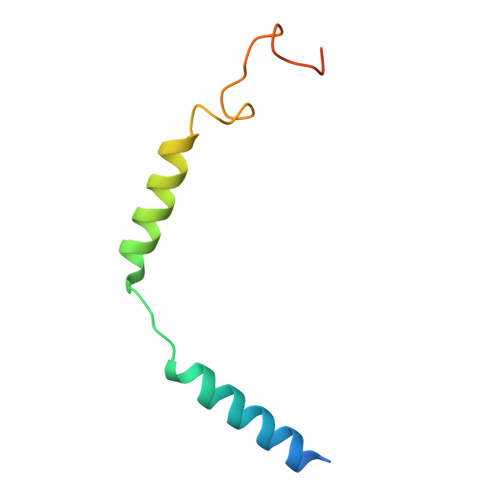
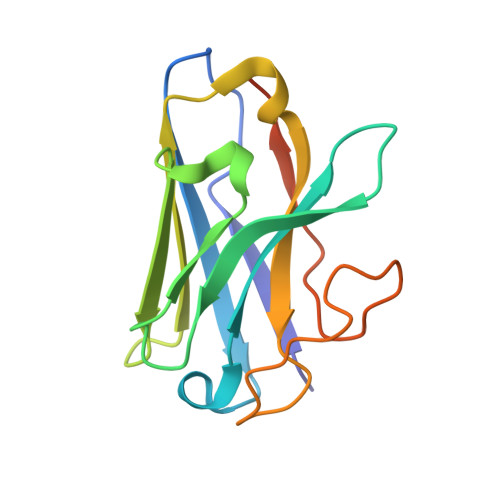
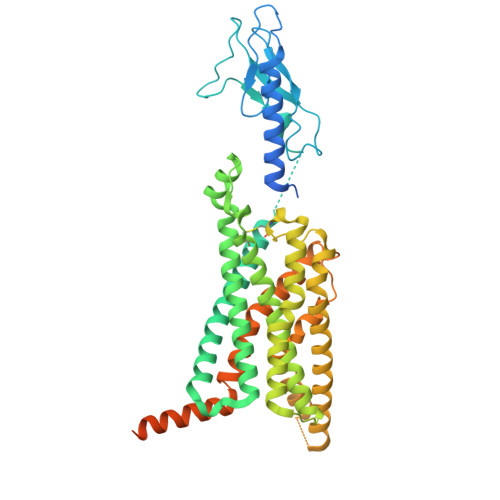
In vivo functional profiling and structural characterization of the human GLP1R A316T variant.
El Eid, L., Manchanda, Y., Austin, G., Deane-Alder, K., Rujan, R.M., Mariam, Z., Oqua, A.I., Belousoff, M.J., de la Serna, J.B., Sloop, K.W., Rutter, G.A., Montoya, A., Withers, D.J., Millership, S.J., Bouzakri, K., Jones, B., Reynolds, C.A., Sexton, P.M., Wootten, D., Deganutti, G., Tomas, A.(2026) Sci Adv 12: eadw0899-eadw0899
- PubMed: 41637494 Search on PubMed
- DOI: https://doi.org/10.1126/sciadv.adw0899
- Primary Citation Related Structures:
9E2A - PubMed Abstract:
Glucagon-like peptide-1 receptor agonists (GLP-1RAs) are effective therapies for type 2 diabetes (T2D) and obesity, yet patient responses are variable, with GLP1R gene variation potentially linked to therapeutic outcomes. A GLP1R natural missense variant, A316T, protects against T2D and cardiovascular disease. Here, we generated and characterized a human GLP1R A316T mouse model. Human GLP1R A316T/A316T mice displayed lower fasting blood glucose versus wild-type littermates even under metabolic stress, as well as slower weight gain and alterations in islet cytoarchitecture, glucagon secretion, and liver metabolism under a high-fat, high-sucrose diet. This was however associated with blunted responses to pharmacological GLP-1RAs in vivo. Further investigations in β cell models demonstrated that human GLP1R A316T exhibits characteristics of constitutive activation but dampened GLP-1RA responses. Results are further supported by cryo-EM analyses and molecular dynamics simulations of GLP-1R A316T structure, collectively demonstrating that the A316T variant governs basal GLP-1R activity and pharmacological responses to GLP-1R-targeting therapies.
- Section of Cell Biology and Functional Genomics, Division of Diabetes, Endocrinology and Metabolism, Department of Metabolism, Digestion and Reproduction, Imperial College London, London, UK.
Organizational Affiliation: