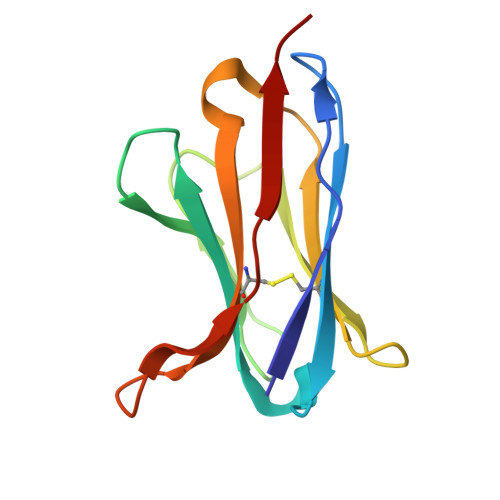
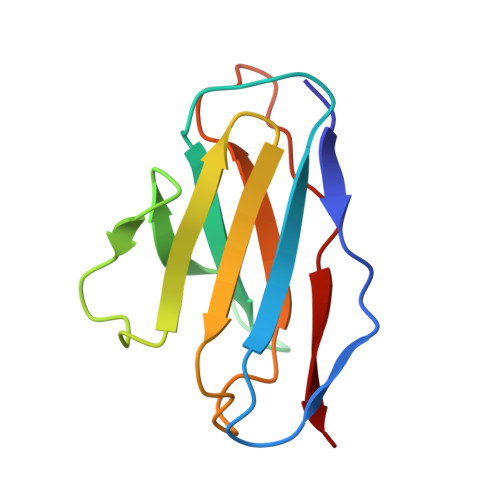
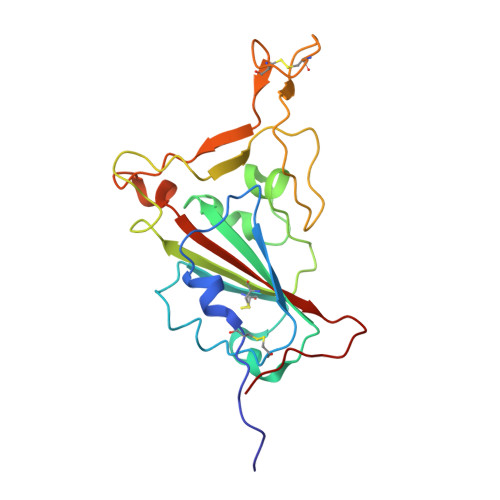
Germinal center-mediated broadening of B cell responses to SARS-CoV-2 booster immunization.
Malladi, S.K., Jaiswal, D., Ying, B., Alsoussi, W.B., Darling, T.L., Dadonaite, B., Civljak, A., Horvath, S.C., Zhou, J.Q., Kim, W., Turner, J.S., Schmitz, A.J., Han, F., Scheaffer, S.M., Farnsworth, C.W., Nachbagauer, R., Nestorova, B., Chalkias, S., Klebert, M.K., Edwards, D.K., Paris, R., Strnad, B.S., Middleton, W.D., O'Halloran, J.A., Presti, R.M., Bloom, J.D., Boon, A.C.M., Diamond, M.S., Bajic, G., Ellebedy, A.H.(2025) Sci Immunol 10: eadu4107-eadu4107
- PubMed: 41071904 Search on PubMed
- DOI: https://doi.org/10.1126/sciimmunol.adu4107
- Primary Citation Related Structures:
9E21 - PubMed Abstract:
Germinal centers (GCs) are key sites for antibody diversification and affinity maturation. SARS-CoV-2 messenger RNA (mRNA) vaccines elicit robust GC B cell responses in humans, but how these responses influence the breadth of immunity against viral variants remains unclear. We analyzed GC B cell responses in nine healthy adults after mRNA booster immunization. We show that 77.8% of the B cell clones in the GC expressed representative monoclonal antibodies (mAbs) recognizing the spike protein, with 37.8% of these targeting the receptor binding domain (RBD). One RBD-targeting mAb, mAb-52, neutralized all tested SARS-CoV-2 strains, including the recent XEC variant. mAb-52 used the IGHV3-66 public clonotype, protected hamsters challenged against the EG.5.1 variant, and targeted the class I/II RBD epitope, closely mimicking the binding footprint of ACE2. Its broad reactivity was driven by extensive somatic hypermutation, underscoring the critical role of GC reactions in shaping cross-variant B cell immunity after SARS-CoV-2 booster vaccination.
- Department of Pathology and Immunology, Washington University School of Medicine, St. Louis, MO, USA.
Organizational Affiliation: