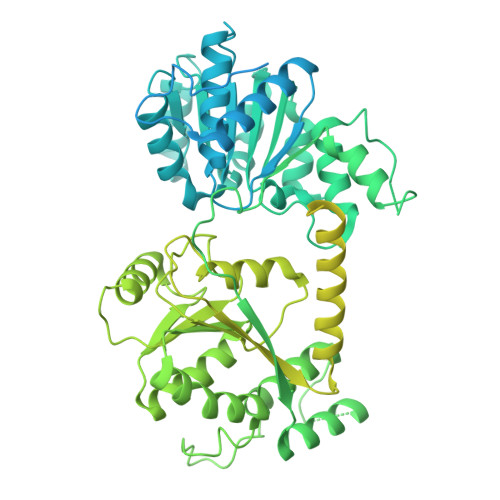
Mechanisms of chromatin remodeling by the human Snf2-type ATPase SNF2H.
Malik, D., Deshmukh, A., Bilokapic, S., Halic, M.(2025) Cell Res 35: 465-468
- PubMed: 40175616 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41422-025-01103-w
- Primary Citation Related Structures:
9E1L, 9E1M, 9E1N, 9E1O, 9E1P, 9E1Q, 9E1R, 9E1U, 9E1V, 9E1W, 9E1X - Department of Structural Biology, St. Jude Children's Research Hospital, Memphis, TN, USA.
Organizational Affiliation: