CO-CRYSTAL STRUCTURE OF CIRCULARLY PERMUTED HUMAN TASPASE-1 BOUND TO LIGAND SMDC994967 1-(ETHENESULFONYL)-4-{[4- (TRIFLUOROMETHOXY)PHENYL]METHYL}PIPERAZINE
Delker, S.L., Edwards, T.E., Abendroth, J.To be published.
Experimental Data Snapshot
Starting Model: other
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Threonine aspartase subunit beta,Threonine aspartase subunit alpha | 339 | Homo sapiens | Mutation(s): 0 Gene Names: TASP1, C20orf13 EC: 3.4.25 | 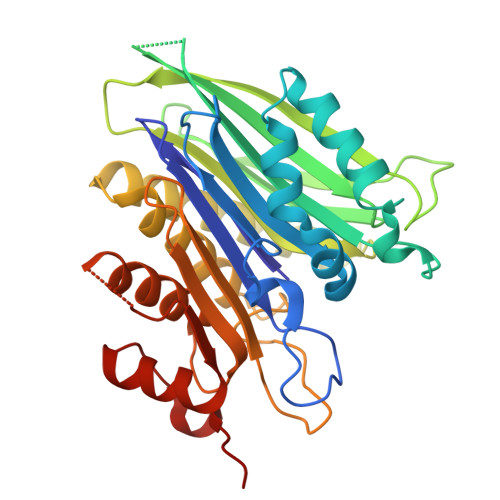 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: Q9H6P5 GTEx: ENSG00000089123 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q9H6P5 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 3 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| R1G (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | F [auth A], I [auth B] | 1-(ethenylsulfonyl)-4-{[4-(trifluoromethoxy)phenyl]methyl}piperazine C14 H17 F3 N2 O3 S GHSHTESCNBFZCN-UHFFFAOYSA-N |  | ||
| CL Download:Ideal Coordinates CCD File | D [auth A], E [auth A], H [auth B] | CHLORIDE ION Cl VEXZGXHMUGYJMC-UHFFFAOYSA-M |  | ||
| NA Download:Ideal Coordinates CCD File | C [auth A], G [auth B] | SODIUM ION Na FKNQFGJONOIPTF-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 60.28 | α = 90 |
| b = 60.28 | β = 90 |
| c = 318.13 | γ = 120 |
| Software Name | Purpose |
|---|---|
| XSCALE | data scaling |
| PHENIX | refinement |
| PDB_EXTRACT | data extraction |
| XDS | data reduction |
| PHENIX | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Institutes of Health/National Cancer Institute (NIH/NCI) | United States | F32GM139242 |