MCM2-7 double hexamer
Zhu, G., Liu, C.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| DNA replication licensing factor MCM4 | A [auth 0], D [auth 4] | 863 | Homo sapiens | Mutation(s): 0 EC: 3.6.4.12 | 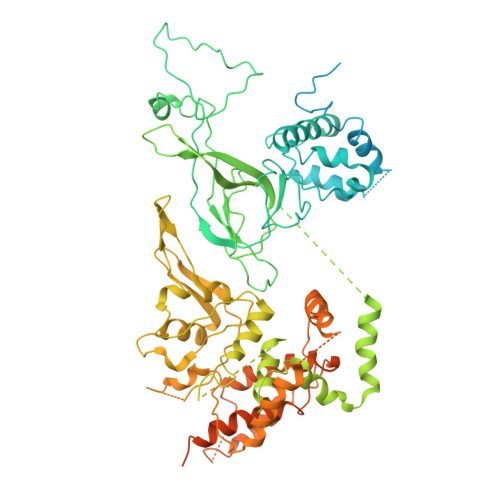 |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P33991 GTEx: ENSG00000104738 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P33991 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| DNA replication licensing factor MCM2 | B [auth 2], H [auth 8] | 904 | Homo sapiens | Mutation(s): 0 EC: 3.6.4.12 | 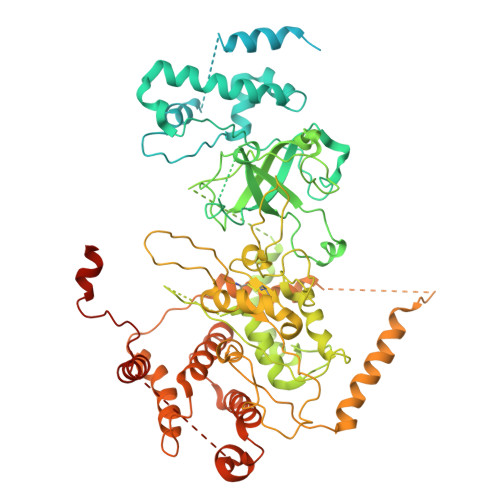 |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P49736 GTEx: ENSG00000073111 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P49736 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 3 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| DNA replication licensing factor MCM3 | C [auth 3], I [auth 9] | 808 | Homo sapiens | Mutation(s): 0 EC: 3.6.4.12 | 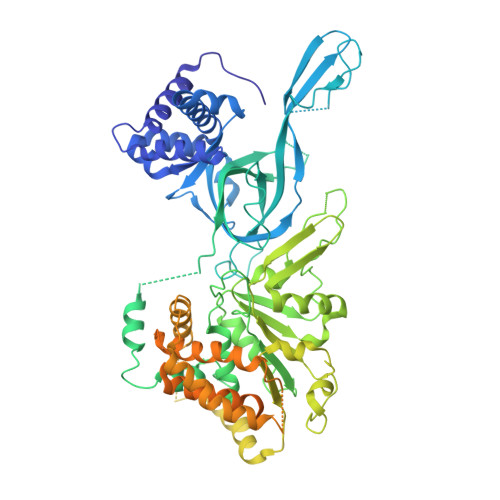 |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P25205 GTEx: ENSG00000112118 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P25205 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 4 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| DNA replication licensing factor MCM5 | E [auth 5], J [auth A] | 734 | Homo sapiens | Mutation(s): 0 EC: 3.6.4.12 | 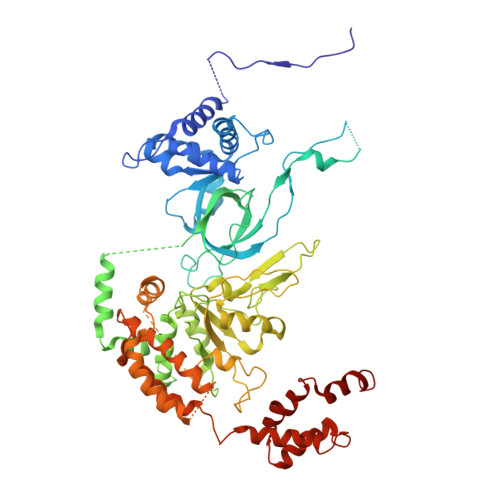 |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P33992 GTEx: ENSG00000100297 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P33992 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 5 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| DNA replication licensing factor MCM6 | F [auth 6], K [auth B] | 821 | Homo sapiens | Mutation(s): 0 EC: 3.6.4.12 | 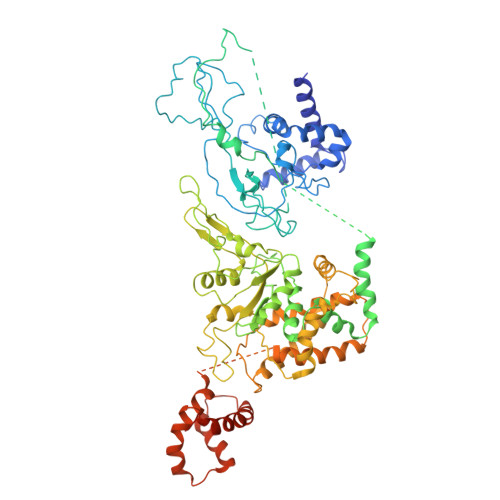 |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: Q14566 GTEx: ENSG00000076003 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q14566 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 6 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| DNA replication licensing factor MCM7 | G [auth 7], L [auth C] | 719 | Homo sapiens | Mutation(s): 0 EC: 3.6.4.12 | 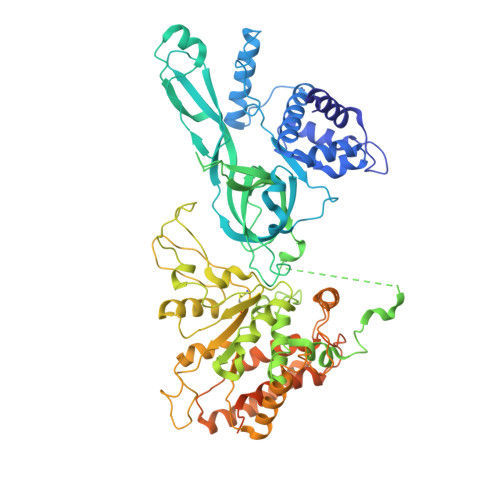 |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P33993 GTEx: ENSG00000166508 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P33993 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Task | Software Package | Version |
|---|---|---|
| MODEL REFINEMENT | PHENIX | |
| Funding Organization | Location | Grant Number |
|---|---|---|
| The University Grants Committee, Research Grants Council (RGC) | Hong Kong | 16101120 |