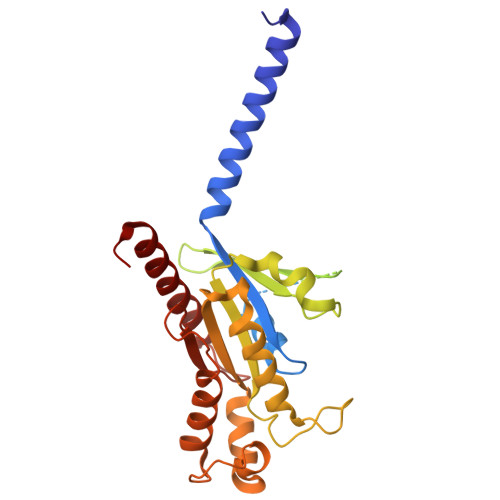
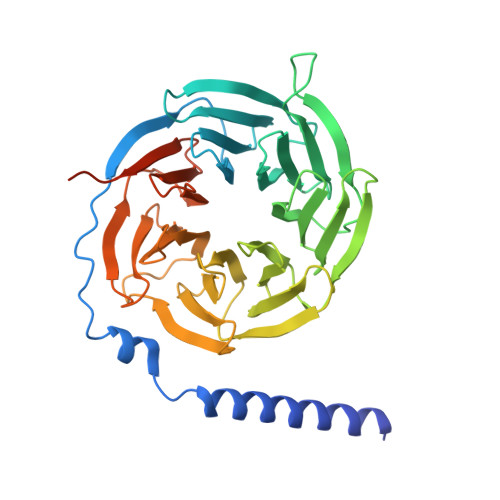
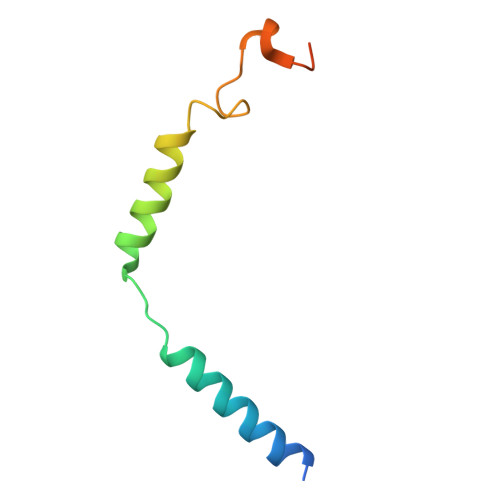
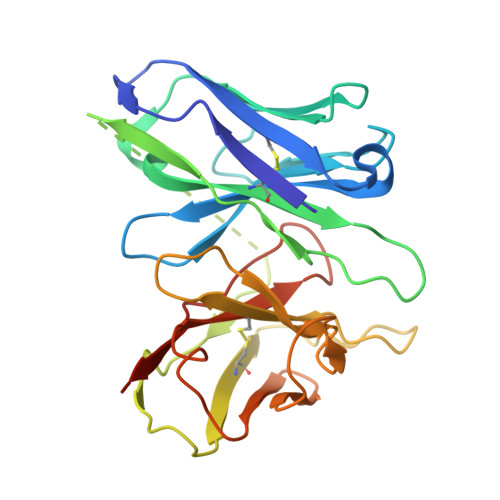
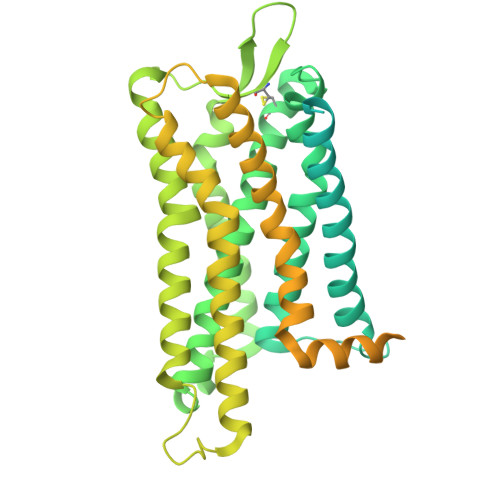
Structural insights into physiological activation and antagonism of melanin-concentrating hormone receptor MCHR1.
Ye, X., Liu, G., Li, X., He, B., Tao, Y., Guan, J., Mu, Y., Liu, H., Gong, W.(2024) Cell Discov 10: 124-124
- PubMed: 39616153 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41421-024-00754-0
- Primary Citation Related Structures:
8WWK, 8WWL, 8WWM, 8WWN, 8YNS, 8YNT - Division of Life Sciences and Medicine, University of Science and Technology of China, Hefei, Anhui, China.
Organizational Affiliation: