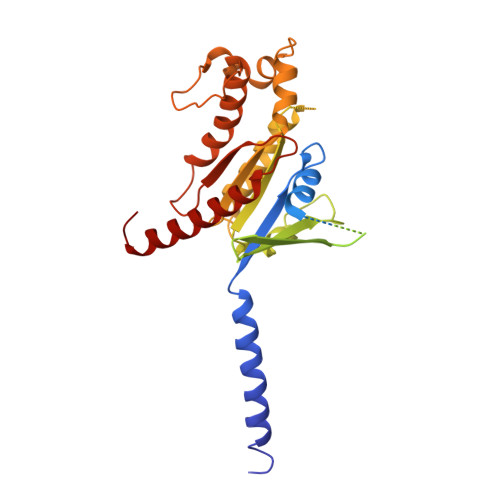
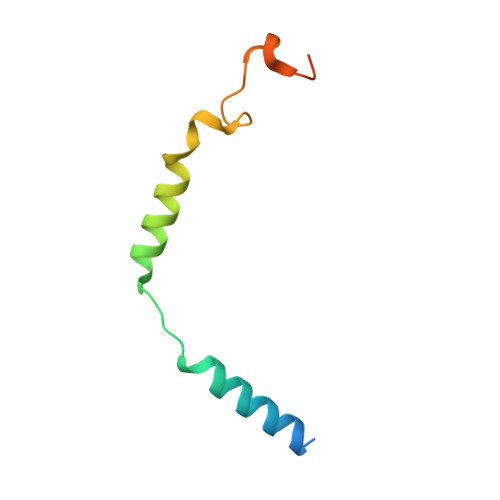
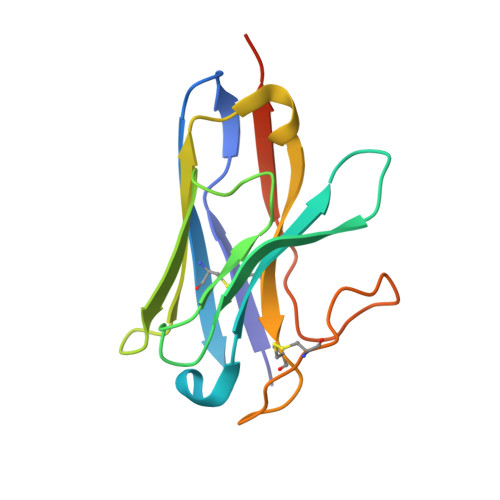
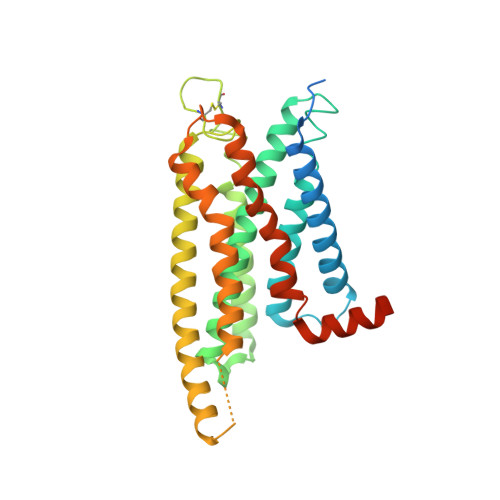
Identification of oleic acid as an endogenous ligand of GPR3.
Xiong, Y., Xu, Z., Li, X., Wang, Y., Zhao, J., Wang, N., Duan, Y., Xia, R., Han, Z., Qian, Y., Liang, J., Zhang, A., Guo, C., Inoue, A., Xia, Y., Chen, Z., He, Y.(2024) Cell Res 34: 232-244
- PubMed: 38287117 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41422-024-00932-5
- Primary Citation Related Structures:
8WW2 - PubMed Abstract:
Although GPR3 plays pivotal roles in both the nervous system and metabolic processes, such as cold-induced thermogenesis, its endogenous ligand remains elusive. Here, by combining structural approach (including cryo-electron microscopy), mass spectrometry analysis, and functional studies, we identify oleic acid (OA) as an endogenous ligand of GPR3. Our study reveals a hydrophobic tunnel within GPR3 that connects the extracellular side of the receptor to the middle of plasma membrane, enabling fatty acids to readily engage the receptor. Functional studies demonstrate that OA triggers downstream G s signaling, whereas lysophospholipids fail to activate the receptor. Moreover, our research reveals that cold stimulation induces the secretion of OA in mice, subsequently activating G s /cAMP/PKA signaling in brown adipose tissue. Notably, brown adipose tissues from Gpr3 knockout mice do not respond to OA during cold stimulation, reinforcing the significance of GPR3 in this process. Finally, we propose a "born to be activated and cold to enhance" model for GPR3 activation. Our study provides a starting framework for the understanding of GPR3 signaling in cold-stimulated thermogenesis.
- HIT Center for Life Sciences, School of Life Science and Technology, Harbin Institute of Technology, Harbin, Heilongjiang, China.
Organizational Affiliation: