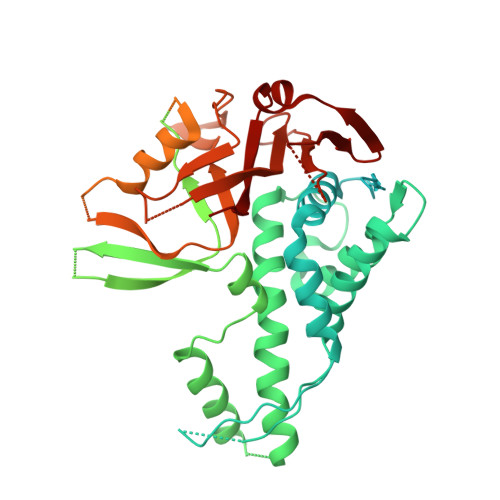
Structural and mechanistic basis for nucleosomal H2AK119 deubiquitination by single-subunit deubiquitinase USP16.
Ai, H., He, Z., Deng, Z., Chu, G.C., Shi, Q., Tong, Z., Li, J.B., Pan, M., Liu, L.(2024) Nat Struct Mol Biol 31: 1745-1755
- PubMed: 38918638 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41594-024-01342-2
- Primary Citation Related Structures:
8WG5 - PubMed Abstract:
Epigenetic regulators have a crucial effect on gene expression based on their manipulation of histone modifications. Histone H2AK119 monoubiquitination (H2AK119Ub), a well-established hallmark in transcription repression, is dynamically regulated by the opposing activities of Polycomb repressive complex 1 (PRC1) and nucleosome deubiquitinases including the primary human USP16 and Polycomb repressive deubiquitinase (PR-DUB) complex. Recently, the catalytic mechanism for the multi-subunit PR-DUB complex has been described, but how the single-subunit USP16 recognizes the H2AK119Ub nucleosome and cleaves the ubiquitin (Ub) remains unknown. Here we report the cryo-EM structure of USP16-H2AK119Ub nucleosome complex, which unveils a fundamentally distinct mode of H2AK119Ub deubiquitination compared to PR-DUB, encompassing the nucleosome recognition pattern independent of the H2A-H2B acidic patch and the conformational heterogeneity in the Ub motif and the histone H2A C-terminal tail. Our work highlights the mechanism diversity of H2AK119Ub deubiquitination and provides a structural framework for understanding the disease-causing mutations of USP16.
- New Cornerstone Science Laboratory, Tsinghua-Peking Joint Center for Life Sciences, Ministry of Education Key Laboratory of Bioorganic Phosphorus Chemistry and Chemical Biology, Center for Synthetic and Systems Biology, Department of Chemistry, Tsinghua University, Beijing, China.
Organizational Affiliation: