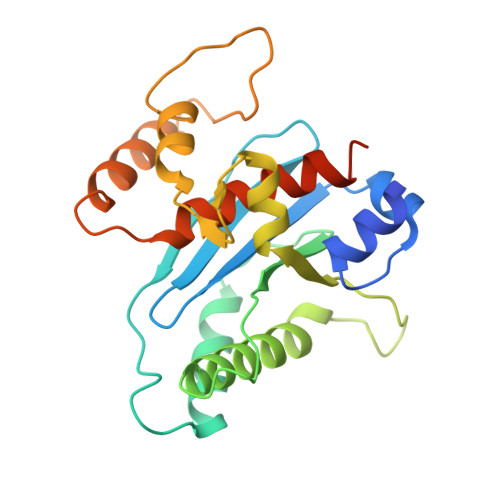
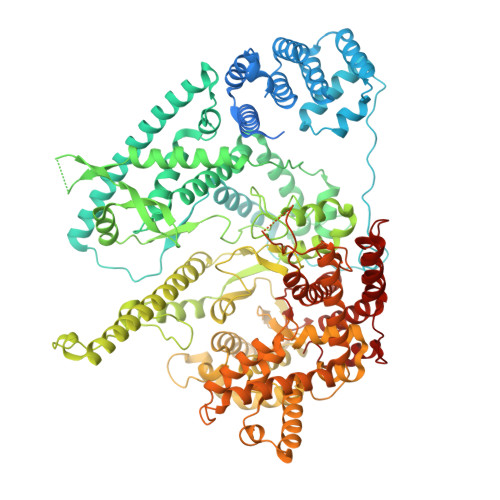
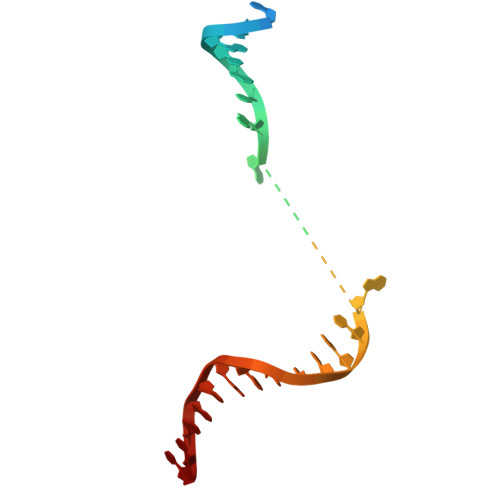
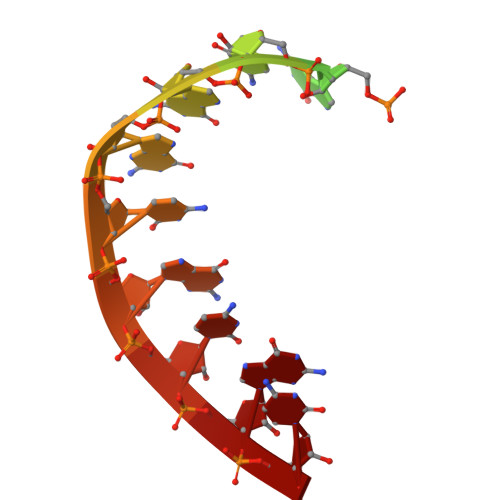
Structural basis for substrate binding and selection by human mitochondrial RNA polymerase.
Herbine, K., Nayak, A.R., Temiakov, D.(2024) Nat Commun 15: 7134-7134
- PubMed: 39164235 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41467-024-50817-9
- Primary Citation Related Structures:
8U8U, 8U8V, 9BDC, 9BDD - PubMed Abstract:
The mechanism by which RNAP selects cognate substrates and discriminates between deoxy and ribonucleotides is of fundamental importance to the fidelity of transcription. Here, we present cryo-EM structures of human mitochondrial transcription elongation complexes that reveal substrate ATP bound in Entry and Insertion Sites. In the Entry Site, the substrate binds along the O helix of the fingers domain of mtRNAP but does not interact with the templating DNA base. Interactions between RNAP and the triphosphate moiety of the NTP in the Entry Site ensure discrimination against nucleosides and their diphosphate and monophosphate derivatives but not against non-cognate rNTPs and dNTPs. Closing of the fingers domain over the catalytic site results in delivery of both the templating DNA base and the substrate into the Insertion Site and recruitment of the catalytic magnesium ions. The cryo-EM data also reveal a conformation adopted by mtRNAP to reject a non-cognate substrate from its active site. Our findings establish a structural basis for substrate binding and suggest a unified mechanism of NTP selection for single-subunit RNAPs.
- Department of Biochemistry and Molecular Biology, Thomas Jefferson University, 1020 Locust St, Philadelphia, PA, 19107, USA.
Organizational Affiliation: