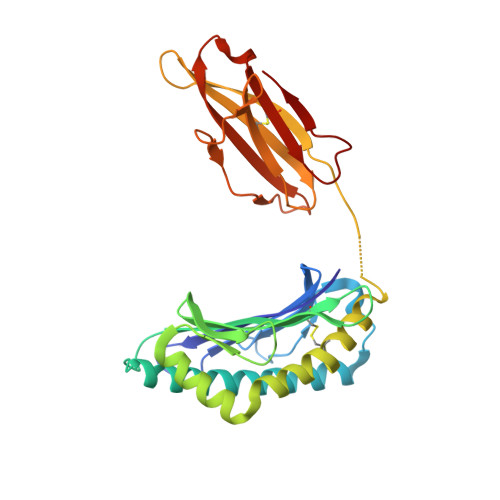
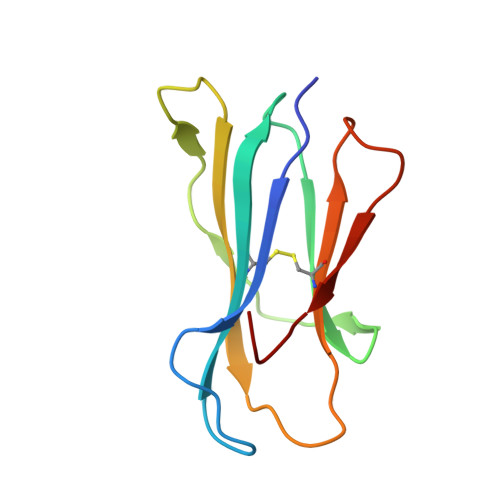
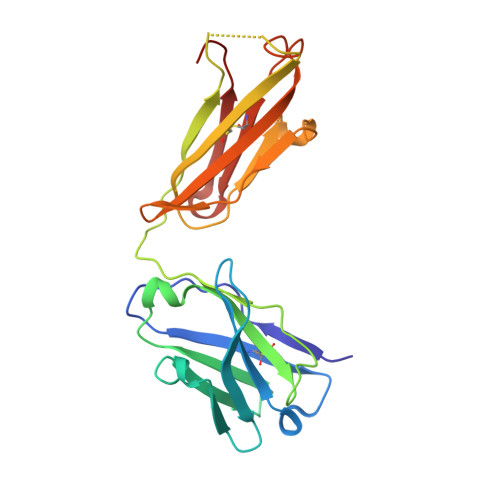
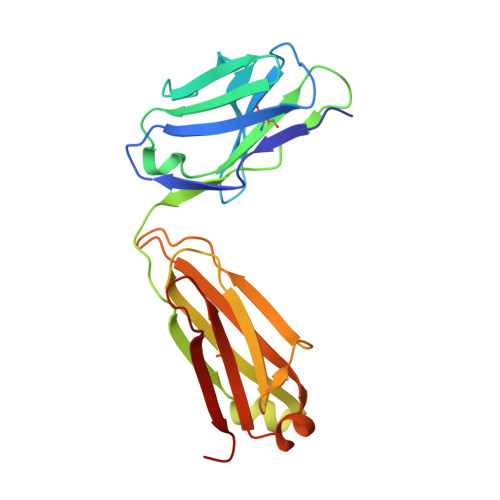
Experimental Structures of Antibody/MHC-I Complexes Reveal Details of Epitopes Overlooked by Computational Prediction.
Boyd, L.F., Jiang, J., Ahmad, J., Natarajan, K., Margulies, D.H.(2024) J Immunol 212: 1366-1380
- PubMed: 38456672 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.4049/jimmunol.2300839
- Primary Citation Related Structures:
8TQ7, 8TQ8, 8TQ9, 8TQA - PubMed Abstract:
mAbs to MHC class I (MHC-I) molecules have proved to be crucial reagents for tissue typing and fundamental studies of immune recognition. To augment our understanding of epitopic sites seen by a set of anti-MHC-I mAb, we determined X-ray crystal structures of four complexes of anti-MHC-I Fabs bound to peptide/MHC-I/β2-microglobulin (pMHC-I). An anti-H2-Dd mAb, two anti-MHC-I α3 domain mAbs, and an anti-β2-microglobulin mAb bind pMHC-I at sites consistent with earlier mutational and functional experiments, and the structures explain allelomorph specificity. Comparison of the experimentally determined structures with computationally derived models using AlphaFold Multimer showed that although predictions of the individual pMHC-I heterodimers were quite acceptable, the computational models failed to properly identify the docking sites of the mAb on pMHC-I. The experimental and predicted structures provide insight into strengths and weaknesses of purely computational approaches and suggest areas that merit additional attention.
- Molecular Biology Section, Laboratory of Immune System Biology, National Institute of Allergy and Infectious Diseases, National Institutes of Health, Bethesda, MD.
Organizational Affiliation: