Characterisation of a novel antimalarial agent targeting haemaglobin digestion that shows cross-species reactivity and excellent in vivo properties.
de Koning-Ward, T.F., Drinkwater, N., Edgar, R.C.S., McGowan, S., Scammells, P.J.(2024) mBio
Experimental Data Snapshot
Starting Model: experimental
View more details
(2024) mBio
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Leucine aminopeptidase | 528 | Plasmodium falciparum | Mutation(s): 3 Gene Names: LAP, PF3D7_1446200 EC: 3.4.11.1 (PDB Primary Data), 3.4.13 (PDB Primary Data) | 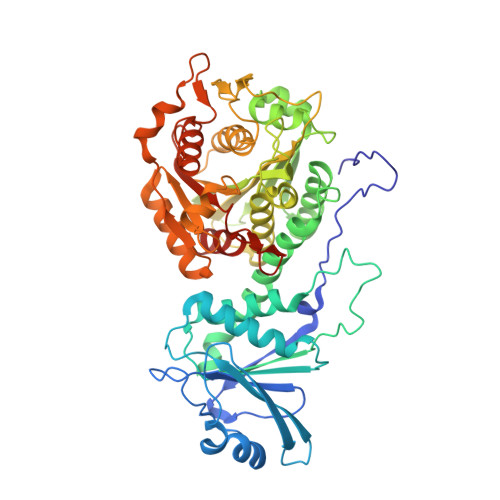 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q8IL11 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 5 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| WRC (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | BB [auth F] EA [auth C] EC [auth J] IB [auth G] LC [auth K] | N-[(1R)-2-(hydroxyamino)-2-oxo-1-(3',4',5'-trifluoro[1,1'-biphenyl]-4-yl)ethyl]-3,3-dimethylbutanamide C20 H21 F3 N2 O3 NRAVUPOEFDEIOQ-GOSISDBHSA-N |  | ||
| 1PE Download:Ideal Coordinates CCD File | AA [auth B] AC [auth I] CB [auth F] DB [auth F] FA [auth C] | PENTAETHYLENE GLYCOL C10 H22 O6 JLFNLZLINWHATN-UHFFFAOYSA-N |  | ||
| SO4 Download:Ideal Coordinates CCD File | EB [auth F] HC [auth J] IA [auth C] JA [auth C] LB [auth G] | SULFATE ION O4 S QAOWNCQODCNURD-UHFFFAOYSA-L |  | ||
| ZN Download:Ideal Coordinates CCD File | AB [auth F] CA [auth C] CC [auth J] DA [auth C] DC [auth J] | ZINC ION Zn PTFCDOFLOPIGGS-UHFFFAOYSA-N |  | ||
| CO3 Download:Ideal Coordinates CCD File | BA [auth C] BC [auth J] FB [auth G] IC [auth K] KA [auth D] | CARBONATE ION C O3 BVKZGUZCCUSVTD-UHFFFAOYSA-L |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 174.204 | α = 90 |
| b = 177.926 | β = 90 |
| c = 229.762 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| Aimless | data scaling |
| PHASER | phasing |
| XDS | data reduction |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Health and Medical Research Council (NHMRC, Australia) | Australia | 1185354 |