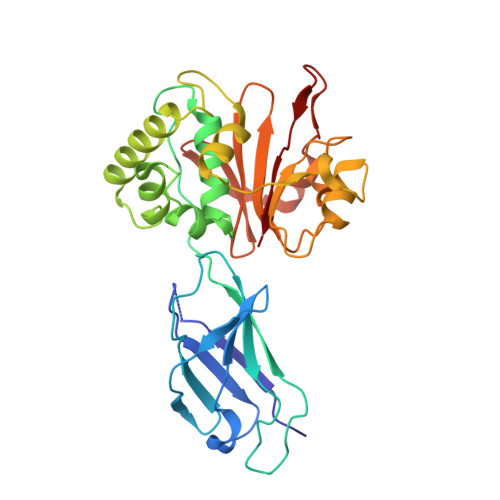
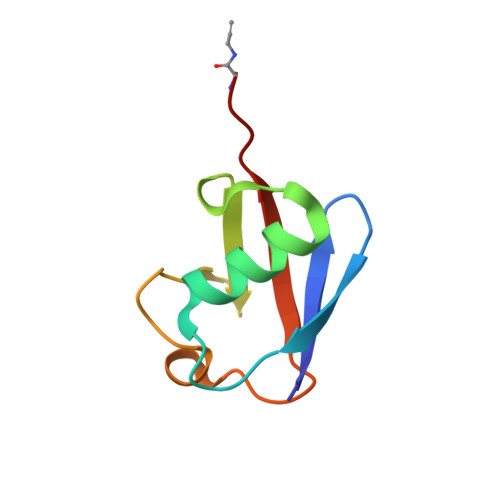
The structural basis for deubiquitination by the fingerless USP-type effector TssM.
Hermanns, T., Uthoff, M., Baumann, U., Hofmann, K.(2024) Life Sci Alliance 7
- PubMed: 38170641 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.26508/lsa.202302422
- Primary Citation Related Structures:
8PZ3, 8Q00 - PubMed Abstract:
Intracellular bacteria are threatened by ubiquitin-mediated autophagy, whenever the bacterial surface or enclosing membrane structures become targets of host ubiquitin ligases. As a countermeasure, many intracellular pathogens encode deubiquitinase (DUB) effectors to keep their surfaces free of ubiquitin. Most bacterial DUBs belong to the OTU or CE-clan families. The betaproteobacteria Burkholderia pseudomallei and Burkholderia mallei , causative agents of melioidosis and glanders, respectively, encode the TssM effector, the only known bacterial DUB belonging to the USP class. TssM is much shorter than typical eukaryotic USP enzymes and lacks the canonical ubiquitin-recognition region. By solving the crystal structures of isolated TssM and its complex with ubiquitin, we found that TssM lacks the entire "Fingers" subdomain of the USP fold. Instead, the TssM family has evolved the functionally analog "Littlefinger" loop, which is located towards the end of the USP domain and recognizes different ubiquitin interfaces than those used by USPs. The structures revealed the presence of an N-terminal immunoglobulin-fold domain, which is able to form a strand-exchange dimer and might mediate TssM localization to the bacterial surface.
- https://ror.org/00rcxh774 Institute for Genetics, University of Cologne, Cologne, Germany t.hermanns@uni-koeln.de.
Organizational Affiliation: