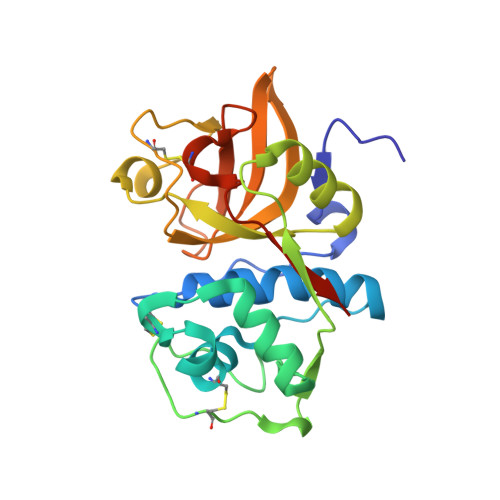
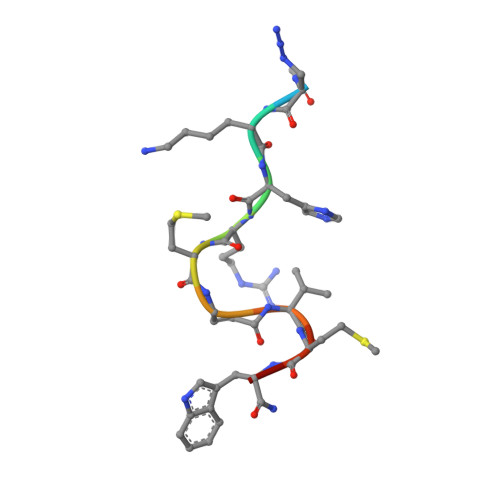
Antibody-peptide conjugates deliver covalent inhibitors blocking oncogenic cathepsins.
Petruzzella, A., Bruand, M., Santamaria-Martinez, A., Katanayeva, N., Reymond, L., Wehrle, S., Georgeon, S., Inel, D., van Dalen, F.J., Viertl, D., Lau, K., Pojer, F., Schottelius, M., Zoete, V., Verdoes, M., Arber, C., Correia, B.E., Oricchio, E.(2024) Nat Chem Biol 20: 1188-1198
- PubMed: 38811854 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41589-024-01627-z
- Primary Citation Related Structures:
8PI3, 8RND - PubMed Abstract:
Cysteine cathepsins are a family of proteases that are relevant therapeutic targets for the treatment of different cancers and other diseases. However, no clinically approved drugs for these proteins exist, as their systemic inhibition can induce deleterious side effects. To address this problem, we developed a modular antibody-based platform for targeted drug delivery by conjugating non-natural peptide inhibitors (NNPIs) to antibodies. NNPIs were functionalized with reactive warheads for covalent inhibition, optimized with deep saturation mutagenesis and conjugated to antibodies to enable cell-type-specific delivery. Our antibody-peptide inhibitor conjugates specifically blocked the activity of cathepsins in different cancer cells, as well as osteoclasts, and showed therapeutic efficacy in vitro and in vivo. Overall, our approach allows for the rapid design of selective cathepsin inhibitors and can be generalized to inhibit a broad class of proteases in cancer and other diseases.
- Swiss Institute for Experimental Cancer Research (ISREC), School of Life Sciences, Swiss Federal Institute of Technology Lausanne (EPFL), Lausanne, Switzerland.
Organizational Affiliation: